Restarting stalled autophagy a potential approach to treating Niemann-Pick disease
(Medical Xpress)—Whitehead Institute researchers have determined that the lipid storage disorder Niemann-Pick type C1 (NPC1) disease is caused not only by defects in cholesterol processing but also in autophagy—a key cellular degradation pathway that malfunctions in many neurodegenerative diseases. Targeting both the cholesterol accumulation and stalled autophagy found in NPC1 with combination therapy could represent a viable treatment strategy.
"Autophagy is a very important mechanism, which contributes to many neurodegenerative diseases and also Niemann-Pick," says Whitehead Founding Member Rudolf Jaenisch, who is a professor of biology at MIT. "Our research indicates that if you improve autophagy, you can somewhat improve the phenotype."
NPC1 is a rare disease caused by an inability to properly metabolize cholesterol and other lipids. Symptoms include learning disabilities, mental retardation, and delayed development of fine motor skills. The disease is fatal, with most children succumbing before age 20, according to the National Niemann-Pick Foundation. Scientists have long explored the problematic accumulation of cellular cholesterol that is a hallmark of the disease, but the research conducted at Whitehead reveals for the first time that impaired autophagy in the cells of NPC1 patients is contributing to disease pathology.
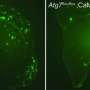
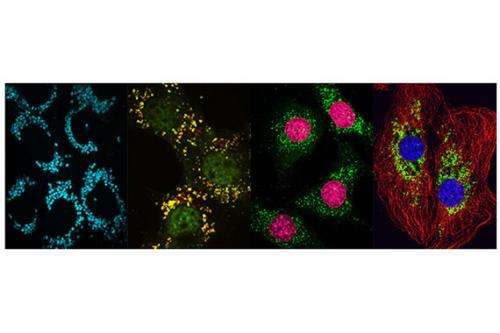
Autophagy is a housekeeping process that allows cells to reuse certain components and prevent the buildup of problematic proteins. Whitehead researchers observed that cells of mice carrying a mutated copy of the human NPC1 gene not only had the characteristic cholesterol accumulations seen in patients, but also abnormalities in the autophagosomes necessary for non-starvation (basal) autophagy. The conclusion: that in NPC1, autophagy is interrupted, causing the process to stall and certain intermediary products of autophagy, including autophagosomes, to amass.
Treatment with HP-beta-cyclodextrin is currently in Phase I clinical trials for improving cholesterol processing in NPC1 patients. However, Whitehead researchers found that high doses of HP-beta-cyclodextrin also impair autophagy and therefore points to careful dosing of this drug. As described in this week's issue of the journal Cell Reports, Whitehead scientists treated diseased cells with a combination of low-dose HP-beta-cyclodextrin, which does not further perturb autophagy, and lithium or rapamycin, which tap into a form of autophagy that is activated by starvation. Together, the drugs rescued cells from both the manifestations of NPC1.
"This dual treatment may have more benefits in patients than using HP-beta-cyclodextrin alone," says Sovan Sarkar, a postdoctoral researcher in Jaenisch's lab and the lead author of the Cell Reports article. "With these drugs together, we can rescue both the cholesterol and autophagy defects. We're not suggesting 'use rapamycin and cyclodextrin'. Since rapamycin and other drugs that inhibit the metabolic mTOR pathway can possibly have side-effects pertaining to suppression of mTOR's critical cellular functions, we're suggesting that we need to stimulate autophagy, ideally by mTOR-independent autophagy activators, and remove cholesterol as a treatment strategy."
Sarkar and others in the Jaenisch lab are currently searching for other potential therapeutics that correct the faulty mechanisms in NPC1. Such drugs may remedy the autophagy issues identified in other neurodegenerative and lipid/lysosomal storage diseases.
More information: "Impaired autophagy in the lipid storage disorder Niemann–Pick type C1 disease" Sovan Sarkar, Bernadette Carroll, Yosef Buganim, Dorothea Maetzel, Alex H.M. Ng, John Cassady, Malkiel Cohen, Souvik Chakraborty, Haoyi Wang, Eric Spooner, Hidde Ploegh, Joerg Gsponer, Viktor I. Korolchuk, Rudolf Jaenisch. Cell Reports, November 27, 2013. DOI: 10.1016/j.celrep.2013.10.042