Advances in muscular dystrophy treatments from targeting endogenous stem cells
In the past few months significant insights into the molecular controls of damaged tissue and how to manipulate them for better regeneration has been revealed for treating rare muscular disorders, which also extends to the larger number of more common degenerative diseases
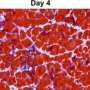
While it is convenient to indicate that the application of stem cells to damaged tissues represents a panacea, the reality is far more dynamic. Following damage the body hosts a series of tightly controlled and interactive events linked to tissue repair, inflammation and removal of redundant tissue which has started to or has already died. These latter steps are performed by a selection of immune cells, macrophages, which following arrival at the damaged site initiate a delicately balanced dance between all the various components at the site of repair. Work performed by the team of Silvia Brunelli at San Raffaele hospital in Milan, and reported in Clinical and Experimental Immunology, has revealed that there is a feed forward loop between these cells and healthy stem cells that are trying to regenerate the tissue. The stem cells themselves actually sustain the macrophages providing key signals to them related to their capacity to repair the tissue; in return the macrophages remain activated clearing away debris while simultaneously providing information to the stem cells themselves to expand.
While happening at the micro level, the mechanism itself represents a key insight and by consequence important pivot through which a tissue can be targetted with therapeutics which provide a comprehensive correction which enables the body to reform a functional tissue following damage.This is a critical aspect of using stem cells to regenerate tissue, as by nature they are not a defined single population of cells, but represent a dynamic collection of interactive and interchanging cell types that give rise to all the functional components of an operational organ. This work has been emphasised by the work of Lorenzo Puri's team from the Fondazione Santa Lucia in Rome and reporting in Genes and Development.
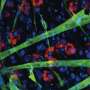
It is becoming apparent that reciprocal interactions between distinct cellular components of the regeneration machinery contribute to establish either a productive or a hostile environment for compensatory regeneration of damaged muscles In particular, the recent identification of multipotent cells residing in the spaces between skeletal muscle, termed Fibroadipogenic progenitors that influence the activity of Muscle Stem cells has revealed an interesting novel cellular contributor to Muscular Dystrophy pathogenesis in particular.
Fibroadipogenic progenitors appear to convert environmental cues into signals that modulate muscle regeneration through functional interactions with Muscle stem cells. For instance, muscle injury-derived signals, such as those from the immune system stimulate Fibroadipogenic progenitors to produce factors that promote Muscle stem cell-mediated regeneration. In contrast, in degenerating muscles such as those in patients suffering from muscular dystrophy at advanced stages of disease, Fibroadipogenic progenitors turn into more differentiated cells, which mediate fat deposition and scar tissue formation thereby disrupting the environment conducive for muscle regeneration. Previous studies showed that pharmacological interventions such as HDACi treatment, which are presently being clinically tested in the Endostem project, can counter the progressive decline in muscle function by promoting regeneration.
Manipulating Fibroadipogenic progenitor function might be a powerful means to alter disease progression; however, the molecular basis for the alternative Fibroadipogenic progenitor phenotypes remains unknown. Prior work had suggested that this diversity in Fibroadipogenic progenitor function in relation to the environmental changes might be under a specific genetic control, which could be influenced by the therapeutics being developed by Endostem.
Puri's team has described a novel molecular network that mediates these same therapeutics ability to reprogram the genetic expression profile of Fibroadipogenic progenitors and support compensatory regeneration at early stages of Muscular dystrophy disease progression.