Predicting the storm: Can computer models improve stem cell transplantation?
Is the human immune system similar to the weather, a seemingly random yet dynamical system that can be modeled based on past conditions to predict future states? Scientists at VCU Massey Cancer Center's award-winning Bone Marrow Transplant (BMT) Program believe it is, and they recently published several studies that support the possibility of using next-generation DNA sequencing and mathematical modeling to not only understand the variability observed in clinical outcomes of stem cell transplantation, but also to provide a theoretical framework to make transplantation a possibility for more patients who do not have a related donor.
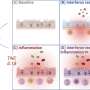
Despite efforts to match patients with genetically similar donors, it is still nearly impossible to predict whether a stem cell transplant recipient will develop potentially fatal graft-versus-host disease (GVHD), a condition where the donor's immune system attacks the recipient's body. Two studies recently published by the online journal Frontiers in Immunology explored data obtained from the whole exome sequencing of nine donor-recipient pairs (DRPs) and found that it could be possible to predict which patients are at greatest risk for developing GVHD and, therefore, in the future tailor immune suppression therapies to possibly improve clinical outcomes. The data provides evidence that the way a patient's immune system rebuilds itself following stem cell transplantation is representative of a dynamical system, a system in which the current state determines what future state will follow.
"The immune system seems chaotic, but that is because there are so many variables involved," says Amir Toor, M.D., member of the Developmental Therapeutics research program at Massey and associate professor in the Division of Hematology, Oncology and Palliative Care at the VCU School of Medicine. "We have found evidence of an underlying order. Using next-generation DNA sequencing technology, it may be possible to account for many of the molecular variables that eventually determine how well a donor's immune system will graft to a patient."
Toor's first study revealed a large and previously unmeasured potential for developing GVHD for which the conventional approach used for matching DRPs does not account. The conventional approach for donor-recipient compatibility determination uses human leucocyte antigen (HLA) testing. HLA refers to the genes that encode for proteins on the surface of cells that are responsible for regulating the immune system. HLA testing seeks to match DRPs who have similar HLA makeup.
Specifically, Toor and his colleagues used whole exome sequencing to examine variation in minor histocompatibility antigens (mHA) of transplant DRPs. These mHA are protein fragments presented on the HLA molecules, which are the receptors on cells' surface to which these fragments of degraded proteins from within a cell bind in order to promote an immune response. Using advanced computer-based analysis, the researchers examined potential interactions between the mHA and HLA and discovered a high level of mHA variation in HLA-matched DRPs that could potentially contribute to GVHD. These findings may help explain why many HLA-matched recipients experience GVHD, but why some HLA-mismatched recipients experience none remains a mystery. This seeming paradox is explained in a companion paper, also published in the journal Frontiers in Immunology. In this manuscript, the team suggests that by inhibiting peptide generation through immunosuppressive therapies in the earliest weeks following stem cell transplantation, antigen presentation to donor T cells could be diminished, which reduces the risk of GVHD as the recipients reconstitute their T-cell repertoire.
Following stem cell transplantation, a patient begins the process of rebuilding their T-cell repertoire. T cells are a family of immune system cells that keep the body healthy by identifying and launching attacks against pathogens such as bacteria, viruses or cancer. T cells have small receptors that recognize antigens. As they encounter foreign antigens, they create thousands of clones that can later be called upon to guard against the specific pathogen that presented the antigen. Over the course of a person's life, they will develop millions of these clonal families, which make up their T-cell repertoire and protect them against the many threats that exist in the environment.
This critical period where the patient rebuilds their immune system was the focus of the researchers' efforts. In previous research, Toor and his colleagues discovered a fractal pattern in the DNA of recipients' T-cell repertoires. Fractals are self-similar patterns that repeat themselves at every scale. Based on their data, the researchers believe that the presentation of minor histocompatability antigens following transplantation helps shape the development of T-cell clonal families. Thus, inhibiting this antigen presentation through immunosuppressive therapies in patients who have high mHA variation can potentially reduce the risk of GVHD by influencing the development of their T-cell repertoire. This is backed by data from clinical studies that show immune suppression soon after transplantation improves outcomes in unrelated DRPs.
The researchers suggest that an equation such as the logistic model of growth, a mathematical formula used to explain population growth, could be employed to predict the evolution of T-cell clones and determine a patient's future risk of GVHD.
"Currently, we rely on population-based outcomes derived from probabilistic studies to determine the best way to perform stem cell transplants. The development of accurate mathematical models that account for the key variables influencing transplant outcomes may allow us to treat patients using a systematic and personalized approach," says Toor. "We plan to keep exploring this concept in hopes that we can tailor the transplantation process to each individual in order to improve outcomes and make transplantation an option for more patients."
More information: The full manuscripts of these studies are available at: journal.frontiersin.org/Journa … .2014.00529/abstract and journal.frontiersin.org/Journa … .2014.00613/abstract