A cell surface protein known to regulate innate immune responses also affects the adaptive immune system
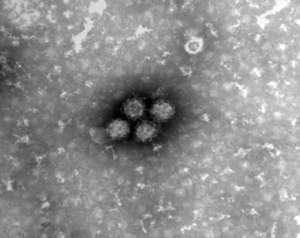
Understanding the immune response triggered by the mosquito-borne Chikungunya virus (CHIKV) is key to developing effective treatments. Now, an international team led by A*STAR researchers has shown that a cell surface protein—the toll-like receptor 3 (TLR3)—plays a critical role in controlling the antibody response to CHIKV, viral replication and pathology.
CHIKV starts with a fever and often debilitating joint pain that can last for weeks to months. First discovered in Tanzania, the virus has spread worldwide—it poses a threat to public health since no specific anti-viral treatment exists.
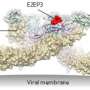
The human body marshals defenses against the virus: the innate immune system kicks in as an immediate response to an infection. Cells express proteins, such as TLR3, that bind viruses like CHIKV (see image) and trigger the expression of cytokines. These cytokines in turn recruit immune cells, like macrophages and neutrophils that destroy the virus.
Lisa Ng from the Singapore Immunology Network and her team sought to clarify the role of TLR3 in CHIKV infection and disease progression.
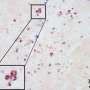
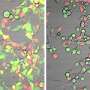
The team showed that in human and mouse fibroblasts, TLR3 inhibits replication of CHIKV. Mice lacking TLR3 had higher levels of viral replication than those with normal TLR3—as more virus entered the blood stream and spread through the body from the infection site. Mice without TLR3 also exhibited more joint inflammation due to higher recruitment of macrophages and neutrophils.
To dissect the role of TLR3 in different cell types, Ng and her team targeted hematopoietic stem cells, the bone marrow cells that give rise to all immune cells. They introduced hematopoietic stem cells that do not express TLR3 into wild-type mice without functional bone marrow. In response to a CHIKV infection, these chimeric mice had higher levels of virus in the blood and took longer to clear the virus.
This delayed clearance pointed to a role of TLR3 in B cell function, cells that produce antibodies in response to a pathogen as part of acquired immunity. "The most surprising finding in our study," says Ng, "is the additional role of TLR3 in adaptive immune response." The team showed that the loss of TLR3 led to antibodies that could no longer neutralize the virus efficiently.
The next step toward effective therapy is to dissect the molecular mechanism that drives virus-mediated inflammation, says Ng. "These data will allow the definition of unique molecular signatures that can be combined with our results to introduce appropriate mechanistic intervention."
More information: "Loss of TLR3 aggravates CHIKV replication and pathology due to an altered virus-specific neutralizing antibody response." EMBO Molecular Medicine 7, 24–41 (2015). dx.doi.org/%2010.15252/emmm.201404459