Investigational personalized cellular therapy tolerated well by patients
Genetically modified versions of patients' own immune cells successfully traveled to tumors they were designed to attack in an early-stage trial for mesothelioma and pancreatic and ovarian cancers at the Perelman School of Medicine at the University of Pennsylvania. The data adds to a growing body of research showing the promise of CAR T cell technology. The interim results will be presented at the American Association for Cancer Research (AACR) Annual Meeting 2015, April 18-22.
"The goal of this phase I trial was to study the safety and feasibility of CART-meso cells in patients with mesothelin-expressing tumors," says Janos L. Tanyi, MD, PhD, an assistant professor of Gynecologic Oncology. "We found no major adverse events associated with the treatment, which suggests that the patients tolerated it very well. But importantly, the T cells successfully targeted the patients' tumor sites and survived in the blood stream for up to 28 days."
Tanyi will present data on five patients (two with ovarian cancer, two with epithelial mesothelioma, and one with pancreatic cancer) who received the new investigational therapy. All patients who received the therapy had cancers which had stopped responding to conventional treatments.
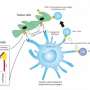
CAR T cells are made from each patient's own immune cells, which have been extracted through apheresis, isolated, and modified to identify and attack tumor cells. In this case, the cells were modified to target cancer cells that express a protein called mesothelin on their surface, so the engineered T cells could identify and kill them.
Though the results suggest the T cells did not attack normal tissues, researchers will follow these patients annually for 15 years in order to more closely observe the CART-meso cells' persistence, and potential antitumor activity.