Unraveling the mystery of a-synuclein in neurodegenerative disease and reversing its course
A team of neuroscientists at Georgetown University Medical Center has shown how a protein, known to accumulate in Parkinson's disease and other neurodegenerative diseases, activates the brain's immune response.
They also demonstrate in their study, published in Science Signaling, that an approved drug used to treat hypertension and a new small molecule compound lessens that immune response.
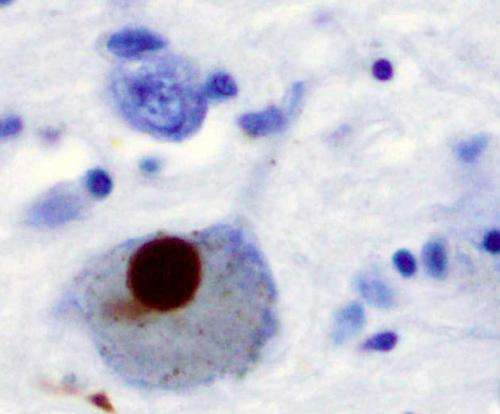
The researchers say there is much work to do to determine how these findings can be translated to human treatment, but the results help uncover some of the molecular mysteries of neurodegenerative diseases characterized by the abnormal accumulation of α-synuclein. The best known of these are Parkinson's and Lewy body dementia. Lewy bodies are large aggregates of α-synuclein mixed with other proteins.
"We have made important progress in understanding how α-synuclein sets up the chronic brain inflammation that is a hallmark of these diseases," says Kathleen Maguire-Zeiss, PhD, associate professor in the department of neuroscience at Georgetown and the study's senior author.
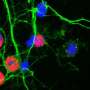
α-synuclein is deeply mysterious because its "normal" function inside neurons, where it is made, is unclear, she says. But it is known that this protein has the ability to rapidly change its structural shape (misfold) making it toxic. Once outside of neurons α-synuclein can activate microglia, cells that act as the main form of immune defense in the central nervous system.
"The real job of microglia is to keep the brain healthy by getting rid of pathogens as well as cellular debris," says Maguire-Zeiss, "However, in a diseased state microglia can become chronically activated, leading to a continuous onslaught of inflammation which is damaging to the brain."
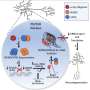
In this study, the Maguire-Zeiss lab found that only a certain size structures of misfolded α-synuclein can activate microglial cells—normal protein and even smaller forms of misfolded α-synuclein cannot. Then the researchers sought to discover precisely how microglia responded to misfolded α-synuclein; that is, which of its many "pattern recognition receptors" reacted to the toxic protein.
Microglia use many different pattern recognition proteins, called toll-like receptors (TLR), to recognize potential threats. The investigators found that misfolded α-synuclein caused TLR1 and TLR2 to come together into one complex (receptor), creating TLR1/2. They traced the entire molecular pathway from the protein's engagement of TLR1/2 at the cell surface to the production of inflammatory molecules.
Then Maguire-Zeiss and her team tested a drug, developed by researchers at the University of Colorado, which specifically targets TLR1/2. They also tested the hypertension drug candesartan, which can target TLR2. Both agents significantly reduced inflammation.
"Use of these drugs provides evidence that we found the right receptors on microglial cells that respond to misfolded α-synuclein," says Maguire-Zeiss. "We think the same process occurs in human brains during disease, but we have much work to do to find this out."