Immune cells interact in surprising ways when responding to skin injuries
When skin is injured or exposed to a pathogen attack, the body's immune system responds rapidly. But the exact skin-cell-based mechanisms behind these responses remain unclear. Now, A*STAR researchers have uncovered how skin-localized cells called dermal dendritic cells (DDCs) respond to sterile skin injuries. Their insights may help to develop new therapeutic interventions administered via the skin, and define the role of skin immune cells in autoimmune and metabolic diseases.
The main job of DDCs is to watch over the skin's micro-environment, and alert the immune system to potential dangers. Upon injury or pathogenic attack, DDCs activate the body's adaptive immune response. Lai Guan Ng and colleagues at the A*STAR Singapore Immunology Network, together with scientists in Australia, wanted to clarify exactly how DDCs are mobilized. They wanted to see how quickly DDCs respond to non-pathogenic inflammation in the form of sterile injury.
"White blood cells, or neutrophils, are able to respond and migrate toward a sterile injury very quickly," says Ng. "We decided to test whether DDCs can do the same. We also hypothesized that neutrophils may be responsible for sending out a signal to DDCs to recruit them to the injury site."
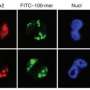
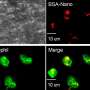
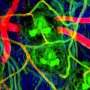
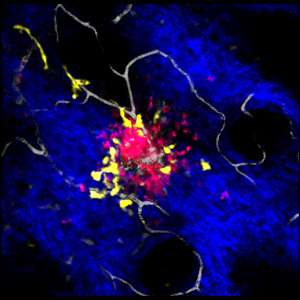
To achieve this goal, the team cross-bred mice with fluorescently labeled neutrophils and DDCs so that both cell types would show up during imaging. They then used a laser to make small, sterile skin lesions on the mice. They followed the responses of the DDCs and neutrophils to the injury site in real-time using a deep tissue imaging technique called intravital multiphoton microscopy (see image).
Their results showed that DDCs migrated in a highly directed manner, gathering around the periphery of the wound approximately 50 minutes after injury (see video). The DDCs appeared to respond to a particular signal to migrate, likely originating from the injury site. However, to the researchers' surprise—although the neutrophils traveled to the injury site within 20 minutes and went straight into the core of the wound—they found that neutrophils had no effect on DDC recruitment.
"When we depleted neutrophils from the mice, DDCs migrated as normal to the injury site," says Ng. "This was a completely unexpected result."
Future experiments will help verify where the DDC signal originates, or indeed whether subsets of DDCs respond to different signals depending on the injury or pathogenic scenario. The team also hopes to extend their research to other disease models to verify the role of DDCs in different medical conditions.
More information: "Real-time imaging of dendritic cell responses to sterile tissue injury." Journal of Investigative Dermatology 135, 1181–1184 (2015). dx.doi.org/10.1038/jid.2014.506