March 5, 2015 report
Immune cells repurpose a stem cell transcription factor as a pathogen sensor
(Medical Xpress)—Among the most abundant of the immune system's first responders, neutrophil granulocytes comprise up to 75 percent of the body's white blood cells. During the acute phase of inflammation, neutrophils migrate to the site of injury or infection through the circulatory system and interstitial tissues to identify and ingest pathogens. They also release proinflammatory cytokines and chemokines which recruit other immune cells to the inflammatory site to eliminate foreign bacteria.
In order to identify pathogens, neutrophils express toll-like receptors (TLRs) for the recognition of bacterial elements. Recently, a group of researchers from the Chinese Academy of Sciences investigating TLRs determined that neutrophils actually use a transcription factor called Sox2, already known for its role in maintaining the self-renewal of ectodermal and endodermal stem cells, as one such TLR. The researchers have published their results in Nature Immunology.
Observing that the morphology of Sox2 present in stem cells resembled that of neutrophils, the researchers determined through immunohistochemical staining that mouse neutrophils, like stem cells, also stained with the antibody for Sox2. But they did not find the Sox2 transcription factor in other cells, including T cells, B cells, natural killer cells, dendritic cells, macrophages, or other granulocytes. "These data indicated that among mature hematopoietic cells, only neutrophils expressed Sox2," the authors write.
The function of Sox2 in immune response
In order to explore the physiological role of Sox2 in the immune system, the researchers bred mice with the conditional deletion of Sox2 in neutrophils; littermates with normal expression of Sox2 served as controls. Both sets of mice were infected with Listeria monocytogenes to determine if Sox2 played an in vivo role in clearing bacteria.
The results were stark, as the Sox2-deficient mice died at a higher rate than their littermate counterparts, and exhibited a bacterial load in the spleen and liver 10,000-fold higher than the control group. This, along with corroborating data, suggest a critical role for Sox2 in neutrophil-mediated bacterial clearance.
Neutrophils require Sox2 to identify bacterial DNA
To test the hypothesis that neutrophils use Sox2 transcription factor as a sensor to identify bacterial DNA, the researchers isolated neutrophils from both sets of mice and transfected the samples in vitro with bacterial genomic DNA. The proinflammatory responses of Sox2-deficient neutrophils were 10 times lower than the unmodified neutrophils.
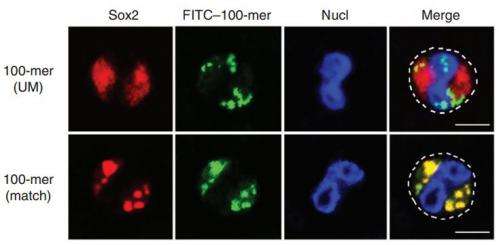
They also explored Sox2 binding sequences in the Listeria genome, determining that 90 percent of sequences that matched Sox2 would bind to it. Furthermore, an analysis of the genomes for other bacteria, including Bartonella, Staphylococcus, and Salmonella found sequences matching Sox2, and which, when transfected into normal neutrophils, produced a proinflammatory response. Unmatched DNA fragments used as controls were unable to bind with Sox2. The authors conclude, "Overall, these results indicated that Sox2 sensed bacterial DNA in neutrophils in a sequence-specific manner."
Furthermore, the researchers determined that, though Sox2 functions as a transcription factor in stem cells, its role in neutrophils is exclusively for the identification of foreign DNA. Though a variety of DNA sensors have been identified, the researchers believe that Sox2 plays a pivotal role in the recognition of pathogens and in distinguishing genetic "self" and "non-self" in order to prevent damage to host cells during immune response. "Thus, our findings have revealed a previously unknown activation pathway in neutrophils and may provide potential therapeutic strategies to augment the innate immunity of neutrophils for the treatment of infectious diseases," the authors conclude.
More information: "Sox2 functions as a sequence-specific DNA sensor in neutrophils to initiate innate immunity against microbial infection." Nature Immunology (2015) DOI: 10.1038/ni.3117
Abstract
Neutrophils express Toll-like receptors (TLRs) for the recognition of conserved bacterial elements to initiate antimicrobial responses. However, whether other cytosolic DNA sensors are expressed by neutrophils remains elusive. Here we found constitutive expression of the transcription factor Sox2 in the cytoplasm of mouse and human neutrophils. Neutrophil-specific Sox2 deficiency exacerbated bacterial infection. Sox2 directly recognized microbial DNA through its high-mobility-group (HMG) domain. Upon challenge with bacterial DNA, Sox2 dimerization was needed to activate a complex of the kinase TAK1 and its binding partner TAB2, which led to activation of the transcription factors NF-κB and AP-1 in neutrophils. Deficiency in TAK1 or TAB2 impaired Sox2-mediated antibacterial immunity. Overall, we reveal a previously unrecognized role for Sox2 as a cytosolic sequence-specific DNA sensor in neutrophils, which might provide potential therapeutic strategies for the treatment of infectious diseases.
© 2015 Medical Xpress