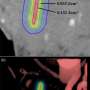
Photodynamic therapy for persistent/recurrent esophageal cancer using Laserphyrin and semiconductor laser
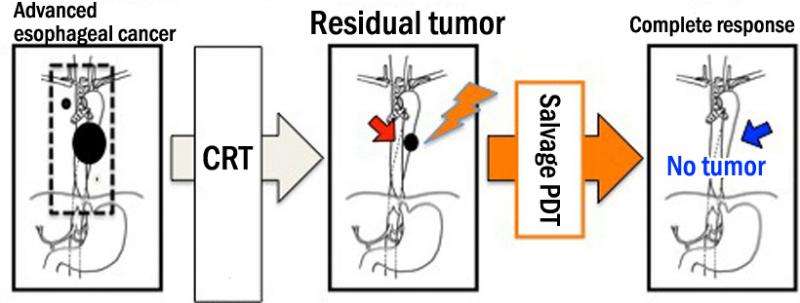
An investigator-initiated clinical trial led by Professor Manabu Muto of the Graduate School of Medicine (involving the seven institutions listed below) has demonstrated that photodynamic therapy (PDT) using Laserphyrin (talaporfin sodium) and semiconductor laser can be effective as a salvage treatment for esophageal cancer patients with local failure after chemoradiotherapy (CRT) or radiotherapy (RT); the procedure achieved a high complete response rate without serious side effects.
Based on this outcome, the photosensitizer, semiconductor laser, and single-use laser probe used in the trial—Meiji Seika Pharma's Laserphyrin 100 mg for Injection and Panasonic Healthcare's PD Laser and EC-PDT Probe—have been approved by Japan's Ministry of Health, Labour, and Welfare for use in the treatment of locally persistent and recurrent esophageal cancer following CRT or RT (an additional indication for the Laserphyrin and the PD Laser, and the first for the EC-PDT Probe). This makes Japan the first country to grant ministerial approval for these three products to be used in salvage treatment.
Having received regulatory approval, the procedure will now be prepared for introduction into hospitals.
PDT is a topical, non-surgical cancer treatment procedure that uses a photosensitizer and a laser to destroy target cells with little impact on the patient's normal tissues and overall health.
While esophageal cancer is treatable and potentially curable with CRT and RT, no standard treatment exists as yet for cases where the tumor has persisted or recurred. In the clinical trial conducted by Professor Muto and others, a staggering 88.5% of patients with persistent or recurred esophageal cancer showed a complete response to PDT.
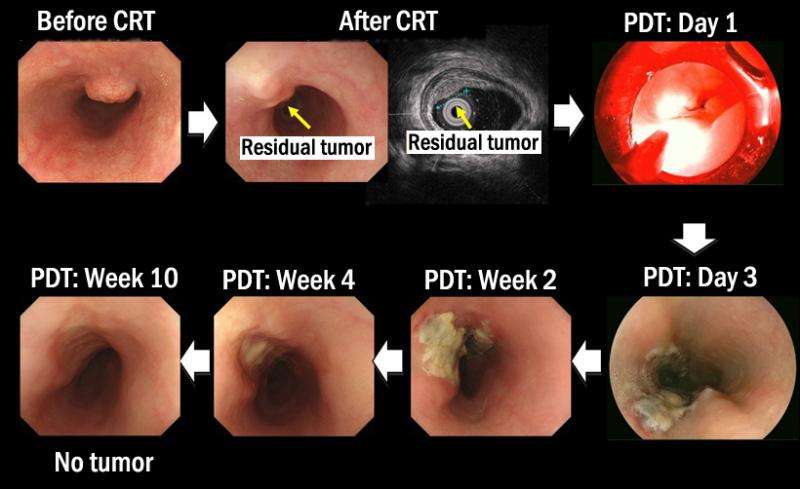
Now approved for clinical use, the procedure—a minimally invasive and curative treatment—is expected to help improve outcomes for patients with similar conditions at hospitals across the country.