September 1, 2015 feature
Take a deep breath: Biodegradable DNA nanoparticles rapidly penetrate mucus barrier for inhaled lung gene therapy
(Medical Xpress)—A number of lung diseases are resistant to, or only marginally handled by, conventional therapies. Thanks to the discovery of numerous genetic targets, gene therapy provides an alternative or complementary therapeutic option. Over the past two decades or more, a large number of gene delivery systems, based on viruses or man-made nanoparticles, have been developed in order to deliver therapeutic nucleic acids to the target cells in the lung, while preventing these cargos from being degraded by the body's protective enzymes before they reach the target. However, while it is readily accessible via inhalation, the mucus lining the lung airways typically traps inhaled foreign matter that is then removed from the lung by being rapidly and continuously swept up towards the larynx to be swallowed into the stomach and degraded. Although this is a critical host defense mechanism, the same airway mucus also traps inhaled therapeutic nanoparticles, such as gene delivery systems, through steric obstruction and/or adhesive interactions, meaning that therapeutic nanoparticles trapped in airway mucus will be rapidly cleared from the lung and so not be able to reach their target cells in the lung. Indeed, several clinically tested viral and non-viral gene delivery systems have been shown unable to efficiently penetrate human airway mucus. In addition, the physiological environment in the lung renders it hard to retain stability of therapeutic nanoparticles until they reach the target cells. Thus, despite over two decades of effort, therapeutically effective lung gene therapy is yet to be realized.
Scientists at the Center for Nanomedicine at Johns Hopkins University School of Medicine, Baltimore have previously shown that dense surface coatings with hydrophilic (readily absorbing or dissolving in water) and uncharged polyethylene glycol (PEG) polymers render the particle surface muco-inert (that is, resistant to being trapped by mucus via adhesive interactions). However, achieving the high PEG densities required for efficient mucus penetration while retaining the stability of gene delivery nanoparticles is challenging. Recently, however, the same researchers developed a simple strategy using a blend of highly PEGylated and non-PEGylated polymers at an optimal ratio to formulate mucus-penetrating DNA nanoparticles (DNA-MPPs) capable of retaining stability in physiological environments as well as rapidly penetrating human airway mucus.
Dr. Jung Soo Suk discussed the paper that he, Prof. Justin Hanes, Dr. Panagiotis Mastorakos and their colleagues published in Proceedings of the National Academy of Sciences. "Non-viral gene delivery systems, being devoid of one or more of shortcomings of virus-based vectors, constitute an attractive alternative for inhaled gene therapy," Suk tells Medical Xpress. (These systems are typically made with natural or synthetic materials possessing a large number of positive charges that interact with negatively charged nucleic acids to form small nanoparticles – a process known as complexation.) "In particular, biodegradable cationic," or positively charged, "polymers provide a superior in vivo safety profile compared to non-biodegradable or slowly degrading systems while providing timely release of nucleic acid payloads that may lead to improved gene delivery efficacy – both features being due to their hydrolytic nature." Hydrolysis is a chemical process of decomposition involving the splitting of a bond and the addition of the hydrogen cation and the hydroxide anion of water.
However, Suk points out, surfaces of these conventionally designed systems are, typically, positively charged, which makes them unlikely not only to retain their colloidal stability in physiological environments, but also to efficiently penetrate negatively charged biological barriers, such as airway mucus, due to the electrostatically-driven adhesive interactions. "Here," he explains, "we engineered a biodegradable polymer-based platform addressing these problems, thereby leading to highly efficient gene transfer to the lung in vivo, surpassing leading non-viral platforms, including a clinically tested system – and perhaps viral vectors as well."
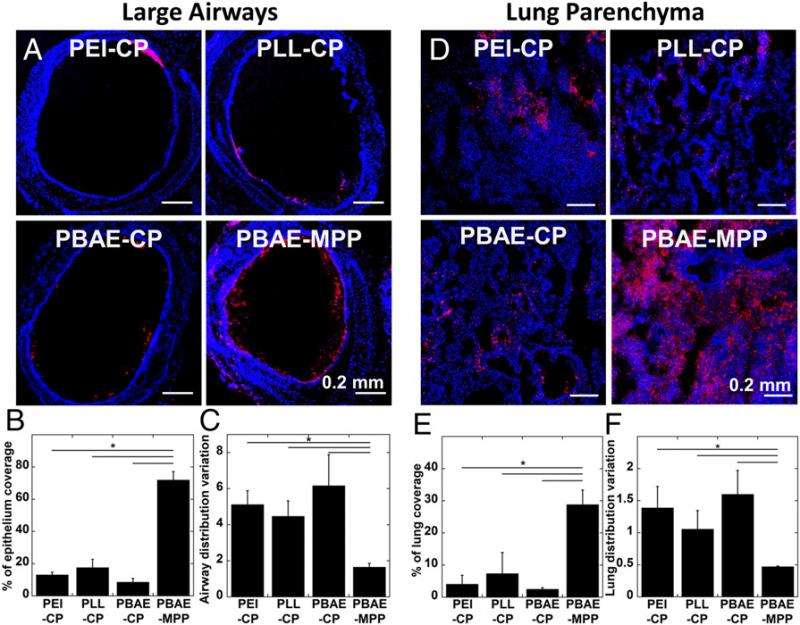
In deciding upon their formulation of highly stable DNA nanoparticles based on biodegradable polymers possessing a dense corona of PEG, Suk notes that they have previously shown that a dense corona of PEG provides efficient penetration of nanoparticles as large as 200 nm in fresh, unperturbed human airway mucus obtained from patients with or without obstructive lung diseases, such as cystic fibrosis (CF) and chronic obstructive pulmonary disease (COPD). Based on these findings, the scientists devised DNA-MPPs – an elegantly simple and highly tunable method of formulating gene delivery nanoparticles capable of efficiently penetrating human airway mucus. The researchers first engineered DNA-MPPs based on gold-standard cationic polymers that have been most widely tested in preclinical settings and clinical trials; more recently, they applied the same approach to poly(b-amino) ester (PBAE) – a state-of-the-art biodegradable polymer.
At the same time, DNA-MPP is simple and generalizable to a variety of biodegradable, bioreducible or other environmentally sensitive cationic polymers. "We can theoretically formulate DNA-MPP based on different cationic polymers with varying polymer structures and/or environmentally sensitive moieties." (A moiety is a part or functional group of a molecule.) "While no single polymer is universally superior to others, the core polymer must be carefully selected depending on the final application, since different polymers possess a repertoire of advantages and disadvantages. In other words, we can feasibly engineer DNA-MPPs with better inner core polymers yet retain the mucus-penetrating property."
The paper describes how the new approach provides robust transgene expression throughout the airways of mouse lungs by overcoming challenging biological barriers. "Based on previous observations, a widespread expression of therapeutic transgene is likely required for therapeutically effective gene therapy of lung diseases," Suk tells Medical Xpress. "However, conventional gene vectors trapped in airway mucus cannot distribute throughout the airway lumen" – that is, pathways of the airways in the lungs. "Here, we show that our DNA-MPPs – regardless of core cationic polymers utilized – provide widespread distribution of gene vectors as well as transgene expression throughout the lung airways."
Suk points out that although cellular or subcellular barriers have been more widely explored, relatively underappreciated extracellular barriers, such as the mucus gel layer, are now recognized as key barriers that must be overcome to achieve therapeutically effective gene therapy in humans. "It should be noted that gene vectors unable to overcome these barriers cannot even reach the target cells, thereby rendering the systems futile."
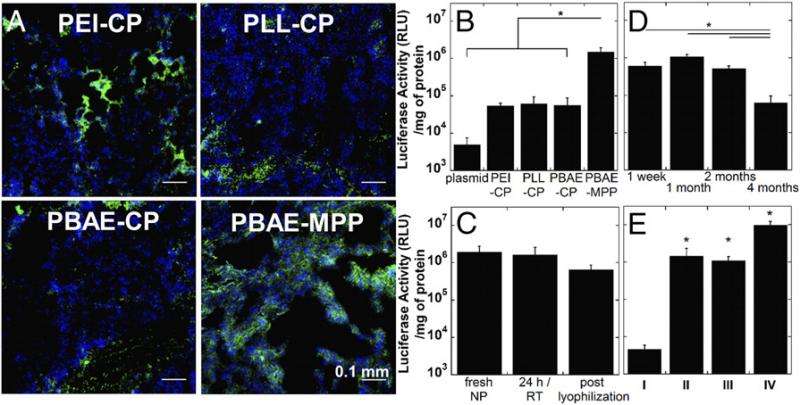
The scientists also showed that their DNA-MPP system provides greater overall transgene expression in vivo compared with gold standard platforms, including a clinically tested system. (Suk adds that since this was a hypothesis-driven study grounded upon carefully designed preliminary mechanistic studies, the scientists expected to see better in vivo performance with the newly designed system – but they could only confirm the hypothesis when the studies were completed.) "It's widely reported that a dense PEGylation undermines the ability of cationic polymer-based gene vectors to transfect cells in vitro," Suk comments. "However, we found that our highly PEGylated system provided significantly greater in vivo gene transfer efficacy compared to gold-standard non-viral gene vectors, including a less PEGylated, clinically tested system – suggesting that the improved lung distribution achieved by DNA-MPPs compared to other systems more than offsets the inferior in vitro gene delivery often observed with densely PEGylated systems." In other words, the study highlights the importance of optimizing gene vectors in vivo rather than in vitro. "I would not say that in vitro optimization is useless," Suk acknowledges, "but in vitro results should be analyzed with caution."
Moreover, the DNA-MPP system demonstrated an excellent safety profile and long-term and consistent transgene expression achieved following a single and repeated administrations, respectively. "One of the primary concerns of non-viral gene delivery systems based on cationic materials, including cationic polymers and lipids, is the potential toxicity originated from their high positive charge densities required for efficient complexation of nucleic acids. Here, we've applied two approaches to address this problem: First, dense PEGylation markedly reduces the positive charges of carrier materials; and second, we utilize biodegradable cationic polymers which will be degraded into small pieces after the therapeutic nucleic acids are delivered to the target cells." In other words, the body will not be exposed to carrier materials with high positive charge densities, which can be cytotoxic, or toxic to cells.
In addition to the current work and their previous demonstration that highly dense surface coatings of hydrophilic and neutrally charged PEG render therapeutic nanoparticles resistant to mucoadhesion, Suk describes other approaches that might enable rapid diffusion in airway mucus. "The mucus barrier can theoretically be tackled by two approaches – namely, by modulating either gene delivery systems or the mucus barrier itself," Suk says. "Our study introduces and demonstrates the first approach, in which we make particles capable of efficiently penetrating the unaltered mucus barrier." Another approach, he explains, involves reducing the barrier properties of airway mucus. "For example, we've previously shown that reducing agents, such as N-acetyl cysteine, can make the pore size of mucus larger; in this case, otherwise impenetrable larger particles can move through the airway mucus. It's also possible to artificially make the mucus gel less adhesive – but these 'mucus-manipulating' approaches must be applied with caution to avoid undermining the essential physiological functions of airway mucus."
As to the scientists' research plans, Suk says that while the studies described in their paper were conducted in healthy inbred mice, they are currently testing their systems in more relevant preclinical disease models. "In general, we're looking into all aspects that could make the use of our DNA-MPPs most efficient potentially in clinics." For example, he illustrates, they are testing system safety after repeated usage and potential batch-to-batch variation, investigating the incorporation of the capability to target specific cells of interest while retaining the mucus-penetrating property; and are optimizing the method of administration.
Regarding the likelihood and timeframe of human trials, Suk tells Medical Xpress that "to be tested in humans, we first need to test our systems in more relevant disease models and confirm the safety in larger animals, such as non-human primates. "While it is hard to exactly gauge the time, we're optimistic because we have top scientists and clinicians in the field as collaborators, and have already initiated efforts to further test our systems."
The newly developed system can be used for treating a range of lung diseases, including CF, COPD, asthma, and lung cancers. "We also recently found that the similarly designed systems provide widespread transgene expression in the brain in vivo following local or systemic administration – and it should be noted that many of brain diseases are characterized by a widely-scattered disease area. We're currently testing these systems in rodent models of brain tumors and Parkinson's disease."
More information: Highly compacted biodegradable DNA nanoparticles capable of overcoming the mucus barrier for inhaled lung gene therapy, Proceedings of the National Academy of Sciences (2015) 112(28): 8720-8725, doi:10.1073/pnas.1502281112
© 2015 Medical Xpress














