New findings in Alzheimers research
Current Alzheimer's research focuses on the amyloid precursor protein (APP), which is responsible for the formation of destructive plaques in the brain. Researchers from Bochum have now demonstrated that APP, in addition to forming those plaques, might also affect the development of Alzheimer's disease via another mechanism.
Protein in the cell membrane
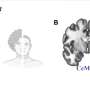
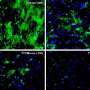
Under certain circumstance, APP causes the formation of spherical structures in the nucleus, so-called spheres. They affect the activity of some genes, which may result in a modulation of neurotransmitter activity. Neurotransmitters are biochemical transmitters that transmit excitation from a nerve cell to other cells. APP itself is a protein that is anchored in the cell membrane. In this capacity, it has several binding sites where other proteins can dock, thus triggering a number of different processes. One of the adaptor proteins is FE65. Under certain circumstances, it can enter the cell nucleus with the aid of the APP. There, FE65 together with other proteins forms the spherical structures specified above. It had previously not been understood in what way they affect the cells.
Comparison between different cell cultures
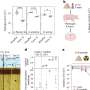
Dr Thorsten Müller from Medizinisches Proteom-Center explains how he and his PhD student Christina Looße have proceeded to gain a better understanding of the spheres' potential influence on the brain: "In our study, we have established a cell culture model inside which we can switch on specific sphere formation. The cells that have been switched on generate spheres." The researchers compared them with cells that were switched off and which did not show any sphere formation. "In the process, we discovered that sphere-forming cells exhibit higher expression of the bestrophin 1-gene," says Müller
Biochemical transmitters and Alzheimer's disease
Bestrophin 1 has recently been described in the context of impaired neurotransmitter activity in Alzheimer's disease. "Elevated levels of the neurotransmitter GABA in the cerebrospinal fluid of Alzheimer's patients have already been described, and our study may shed light on how neurotransmitter modulations correlate with the APP," as Dr Thorsten Mülller elaborates the relevance of his research work for medical science.
Starting point for future therapies
Contrary to previous assumptions, the APP could thus affect the development of Alzheimer's disease, inasmuch as it affects an inhibition of neurotransmitter activity and is not relevant as a precursor of plaques in the first place. "This hypothesis might provide interesting starting points for the development of therapies for the treatment of Alzheimer's disease," says Thorsten Müller.
More information: Christina Loosse et al. Nuclear spheres modulate the expression of BEST1 and GADD45G, Cellular Signalling (2016). DOI: 10.1016/j.cellsig.2015.10.019