Researchers unveil architecture of mitochondrial calcium uniporter
Mitochondria are key integrators of cellular calcium (Ca2+) signaling and energy metabolism. Previous studies demonstrated that isolated mitochondria could buffer huge amounts of Ca2+ via a highly selective channel called the uniporter. Uptake of Ca2+ via the uniporter is known to activate the citric acid cycle, while its overload leads to cell death. The uniporter has been studied extensively for over 50 years, but its molecular identity remained elusive until in 2011, when computational genomics studies discovered its molecular components. The centerpiece of the uniporter, the calcium-conducting subunit, is the mitochondrial calcium uniporter (MCU). MCU exhibits the unique property of both high selectivity and high conductance for calcium, which makes it an intriguing structural target being pursued by many structural biology labs worldwide.
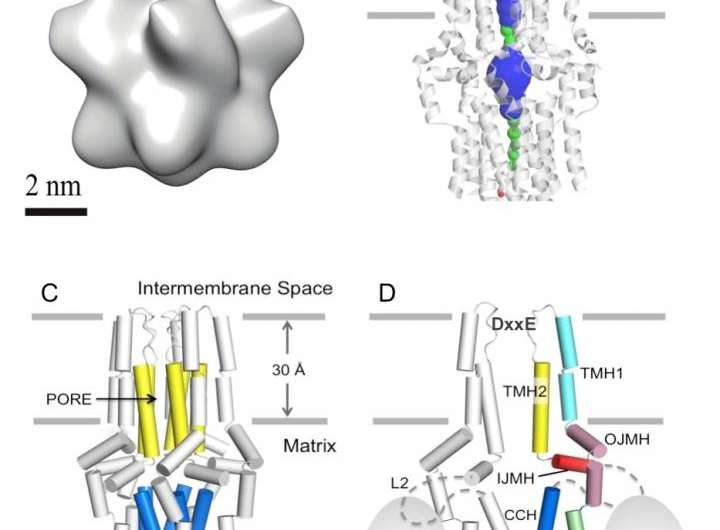
Prof. James J. Chou's lab, together with collaborators, revealed the MCU architecture using nuclear magnetic resonance (NMR) and electron microscopy (EM). The MCU structure is a homopentamer with the second transmembrane helix forming a hydrophilic pore across the membrane. The critical DxxE amino acid sequence motif forms the pore entrance featuring two carboxylate rings, which appear to be the ion selectivity filter. The structure represents a novel ion channel architecture and suggests a passage for calcium transport. This is one of the largest structures characterized by NMR, providing a structural blueprint for understanding the function of this channel.
In addition to MCU, the uniporter complex also includes other components, including the channel gate keeper EMRE (essential MCU regulator), and regulatory subunits MICU1 (mitochondrial calcium uptake 1), MICU2 (mitochondrial calcium uptake 2), and MCUb (MCU isoform b). The complexity implies intricate regulation of MCU, which appears to be an ancient calcium channel that is a part of the earliest eukaryotes. The reported MCU pore architecture represents the first step toward understanding how this complex calcium uniporter works.