Toxic peptide payload can be delivered exclusively to cancer cells
A drug that can kill cancer cells while leaving normal cells unharmed may be within our grasp thanks to research from A*STAR, although the approach is still several years away from clinical trials.
The discovery began when Sheng-Hao Chao's team at the A*STAR Bioprocessing Technology Institute realized that a segment of the hexamethylene bisacetamide-inducible protein 1 (HEXIM1) they were studying, known as the basic region (BR) peptide, was similar to a key region of the tumor-suppressing protein p53. Activation of p53 in damaged cells either leads to recovery of the cell via DNA repair or initiation of a cell-death process known as apoptosis. Since HEXIM1 was known to interact with p53 and other cancer-related proteins, the researchers speculated that BR might be involved in the p53 pathway.
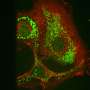
To test this, the team engineered a new protein by connecting a cell-penetrating peptide to the BR peptide. While the BR peptide could not enter cells alone, the combined protein was readily taken up and proved toxic to the cells. By attaching a signal to target breast cancer cells, the team ensured that BR was delivered exclusively to these cells. The BR complex efficiently eliminated breast cancer cells but left normal cells unharmed. "Our peptide acts like a 'professional killer,' killing only the targeted cancer cells," says Chao. "This unique feature makes it a safer choice as a toxic payload for targeted therapies against cancerous cells."
The BR peptide can also be combined with different molecules, such as antibodies, to target other cancer cell types. Its ability to selectively kill cells is a major improvement over existing toxic peptides, which can enter cells without assistance and therefore cause unwanted side-effects by killing normal cells.
Cancers sometimes overcome p53's suppressive action by regulating apoptosis or even p53 itself, but this will not be possible with BR. The team found that treatment with BR killed breast cancer cells within minutes, meaning that they weren't undergoing apoptosis, which takes hours. Cells lacking p53 were also killed, and further experiments demonstrated that BR acts not via the p53 pathway but through another protein, nucleophosmin, which is essential for cell growth and survival.
"That was totally unexpected," says Chao. "The combination of a unique safety feature and unique killing mechanism could make the BR peptide very attractive for developing new therapeutics against cancers. That's what we really hope."
Chao's team is currently engineering peptides with BR connected to other cancer-targeting molecules, as well as testing the peptide in mice.
More information: Neo, S. H., Lew, Q. J., Koh, S. M., Zheng, L., Bi, X. & Chao, S.-H. Use of a novel cytotoxic HEXIM1 peptide in the directed breast cancer therapy. Oncotarget 7, 5483–5494 (2015). DOI: 10.18632/oncotarget.6794