Impaired recycling of mitochondria in autism?
Tuberous sclerosis complex (TSC), a genetic disorder that causes autism in about half of those affected, could stem from a defect in a basic system cells use to recycle their mitochondria, report scientists at Boston Children's Hospital. The scientists believe their findings, published online October 18 by Cell Reports, open new treatment possibilities not just for TSC, but possibly for other forms of autism and some neurologic disorders.
Mitochondria, the organelles responsible for cellular energy production and metabolism, constantly get recycled. Through a process known as autophagy ("self-eating"), cells literally digest their damaged or aging mitochondria, clearing the way for healthy replacements. (Research on how autophagy works earned a Nobel Prize earlier this month.)
The new study, led by Mustafa Sahin, MD, PhD, and co-first authors Darius Ebrahimi-Fakhari, MD, PhD, a resident at Boston Children's Hospital, and medical student Afshin Saffari, in Boston Children's F.M. Kirby Neurobiology Center, shows that autophagy is defective in TSC. The scientists further showed that two existing classes of drugs counter the defect: the epilepsy drug carbamazepine, and drugs known as mTOR inhibitors. When treated, the dysfunctional neurons were able to clear damaged mitochondria and replenish healthy mitochondria, restoring a normal turnover.
"Our findings point to possible treatments for enhancing mitophagy for some neurodevelopmental and neurodegenerative diseases," says Sahin, who is also director of the Translational Neuroscience Center at Boston Children's and senior author on the paper.
Out with the old, in with the new
Defects in mitophagy, or autophagy of mitochondria, have already been implicated in a number of neurologic disorders such as Parkinson's disease and Alzheimer's disease. Mitochondria have also been studied in autism for years, but the findings have been largely anecdotal and inconclusive, in part because the autism population is diverse and hard to define.
"We decided to use tuberous sclerosis, a genetically defined disorder that has a high incidence of autism, as a model to understand the role of mitochondrial dynamics," says Sahin.
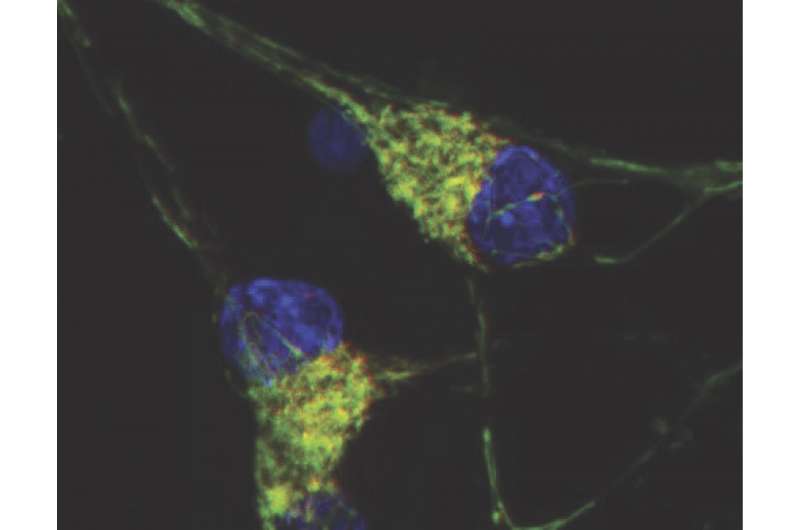
Sahin, Ebrahimi-Fakhari and colleagues studied both rat neurons and patient-derived neurons (created from induced pluripotent stem cells) affected by TSC and used live-cell imaging to examine the distribution and dynamics of mitochondria. They found that the TSC neurons as a whole had more mitochondria, and in particular more fragmented and dysfunctional mitochondria.
Axons take the hit
Then they examined the neurons' axons, the projections that send messages to other cells. Mitochondria play a critical role in axons, and are found in high numbers at presynaptic sites—the tips of axons that form synapses or junctions with other neurons and release neurotransmitters. But the axons of both rat neurons and neurons from TSC patients were depleted of mitochondria.
"We think this could have implications for how neurons talk to each other," says Ebrahimi-Fakhari. "Synapses that lose the support of mitochondria might be releasing neurotransmitters too much or too little."
Diving deeper, they found that while mitophagy was increased in the body of the cell, it was reduced in the axons. Although proteins involved in the early steps of mitophagy increased in the axons, autophagosomes and lysosomes—the organelles that do the digesting—failed to appear around the damaged mitochondria. Instead, the mitochondria were being shuttled out of the axons, back to the body of the cell, without being replaced.
Therapeutic opportunity?
The researchers were able to restore normal mitophagy and replenish functioning mitochondria—in both neurons in a dish and in live mice—in several ways:
- by reintroducing a healthy copy of the gene mutated in TSC
- by adding rapamycin, an mTOR inhibitor that the Sahin lab has shown to improve TSC in animal models and that is currently in clinical trials
- with carbamazepine, a common anti-seizure medication, that enhances autophagy through a different mechanism of action than mTOR inhibitors.
Most notably, mitochondria were replenished at presynaptic sites, where their presence is most critical.
The findings shed intriguing light on what is already known about TSC and autism. Growing evidence, including from previous studies in the Sahin lab, shows that autism, intellectual disability and seizures in many patients with TSC may result, at least in part, from synaptic dysfunction. Autism itself is increasingly seen as a disorder of synapses—and this study hints at one possible way synapses might go awry.
"Our work defines mitochondrial homeostasis as a therapeutic target for TSC, and may also have implications for other neurological diseases that involve mitochondrial dysfunction," says Ebrahimi-Fakhari.