Cancer cells hijack healthy cells to help them spread to other organs
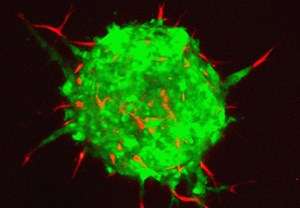
An interaction between two proteins enables cancer cells to use the physical forces of healthy cells to start spreading to other parts of the body.
The finding by researchers from the Francis Crick Institute in London and the Institute for Bioengineering of Catalonia (IBEC) in Barcelona is published in the journal Nature Cell Biology.
The process by which cancer cells separate from the original tumour to form new tumours in other organs or tissues of the body is called metastasis, and it is responsible for the majority of deaths in patients with cancer.
Many cancers initially have a very limited ability to invade the tissue around them. But over time they find the mechanism to do so, increasing the aggressiveness of the cancer.
The British and Spanish research team identified a mechanism by which cancer cells manage to escape the tumour to promote metastasis. They revealed that tumour cells can reprogramme their healthy neighbours, dragging them out of the tumour and into other tissues.
The victims of this 'kidnapping' are fibroblasts, a cell type that is responsible for the synthesis and organization of the extracellular matrix - the scaffolding in which cells reside and which helps give structure to tissues in the body.
"Fibroblasts are professionals in maintaining healthy tissues, and are able to create tunnels in the tissues to travel through," says Anna Labernadie of IBEC and first author of the study. "They use this ability to restore the tissues when we suffer an injury."
The researchers, led by Xavier Trepat at IBEC and Erik Sahai at the Crick, showed that cancer cells use the mobility of fibroblasts to escape the tumour and travel through tissues. The surrounding tumour, or stroma, modifies the fibroblasts and uses them for the invasion into other tissues. From this point, the fibroblasts start to serve the cancerous cells, tracing paths for them through the extracellular matrix surrounding the tumour.
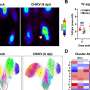
The scientists have now shown that fibroblasts not only create tunnels beyond the stroma, but exert physical forces to drag cancer cells through these tunnels, promoting the invasion of healthy tissue.
"It's like a train: the fibroblasts are locomotives and the cancer cells are wagons," says Labernadie.
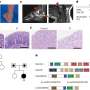
The group were able to identify the mechanism by which cancer cells stick to fibroblasts and move together. It is down to an interaction between two different proteins, one located on the surface of cancer cells called E-cadherin, and another expressed on the surface of fibroblasts, called N-cadherin.
"These proteins are the nanoscale hooks that enable cells to join together," says Xavier Trepat of IBEC. "Until now, we knew very little about this interaction.
The researchers used cells and tissues obtained from patients with lung and skin cancer. "We identified this mechanism in very different tumours, which suggests that it may be universal," stresses Xavier. "Because of the poor prognosis for patients who have suffered cancer metastasis, identifying the mechanisms that promote tumour growth can help us find ways to stop it."
The paper, A mechanically active heterotypic E-cadherin/N-cadherin adhesion enables fibroblasts to drive cancer cell invasion, is published in Nature Cell Biology.
More information: Anna Labernadie et al. A mechanically active heterotypic E-cadherin/N-cadherin adhesion enables fibroblasts to drive cancer cell invasion, Nature Cell Biology (2017). DOI: 10.1038/ncb3478