Chemical stimuli can support growing of stable cartilage cells
Cell-based therapies could offer a way to treat cartilage injuries before the ultimate damage of osteoarthritis on articular cartilage. Cartilage-derived chondrocytes can be used for cartilage repair, but the expanded cells quickly lose their cartilage-specific characteristics. Researchers at Umeå University in Sweden show that a cytoskeleton-affecting chemical stimuli by a small molecule can be used to enhance the production of cartilage-specific molecules.
"Cartilage cells (or chondrocytes) isolated and expanded from cartilage tissue have great potential as a cell-based therapy for patients with, for example, traumatic cartilage defects," says Juha Piltti, doctoral student at the Department of Integrative Medical Biology.
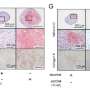
"The development of novel treatments demands both a good knowledge of articular cartilage biology and tissue engineering methods. A key challenge in this has been to stabilize the phenotype and production of extensive extracellular matrix by the grown cartilage cells so that they can be useful in cell-based therapy. Human chondrosarcoma HCS-2/8 cells, which are a type of benign bone cancer cells, can be used to investigate how optimization of culture conditions could improve the synthesis of cartilage-specific molecules."
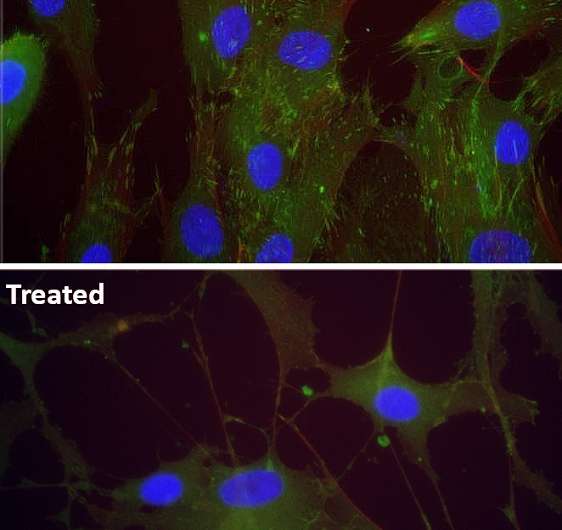
In his research, Juha Piltti has focused on a small molecule, which causes Rho-kinase inhibition and regulation of specific down-stream protein pathways involved in many physiological processes. The molecule has the potential to promote and support extracellular matrix production in cultured chondrocytes. The research has also investigated the role of Rho-kinase inhibition on cellular-scale protein expression and stimulation of cellular differentiation of fibroblast cells.
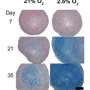
The results showed that, when applied, a long-term inhibition of Rho-kinase signaling increased the expressions of chondrocyte-specific genes and differentiation markers in human chondrosarcoma 2/8 cells. The Rho-kinase inhibition stimuli under low oxygen conditions (of 5 percent oxygen atmosphere), which is a condition corresponding to conditions in the body's cartilage, produced a more effective increase in chondrocyte-specific gene expression and synthesis of extracellular matrix components by HCS-2/8 cells.
"This kind of markers can be useful to monitor responses during chondrocyte in vitro culturing, but also be applicable to monitor status of the chondrocytes during the different kinds of cartilage therapies. My hope is that these conditions can be used to enhance extracellular matrix production," says Juha Piltti.
According to the researcher, further studies are warranted to confirm responses in primary cells and to reveal whether Rho-kinase inhibition stimuli can be used to manufacture a better cartilage-type of tissue in laboratory. Previously, it has been suggested that S100A1 and S100B proteins can act as markers of chondrocyte differentiation status, although the concept is still not commonly adopted so far.
"My studies show a correlation between extracellular matrix production and the specific S100A1 and S100B proteins synthesis. This finding offers new evidence of the applicability of S100A1 and S100B as markers of chondrocyte phenotype," says Juha Piltti.
More information: Responses of fibroblasts and chondrosarcoma cells to mechanical and chemical stimuli. umu.diva-portal.org/smash/record.jsf?pid=diva2%3A1089444&dswid=5873