Pain neuron may protect fungal inflammation and bone destruction
Scientists have discovered unexpected functions of pain neurons, finding that they inhibit fungi-induced osteo-inflammation via the CGRP-Jdp2 axis.
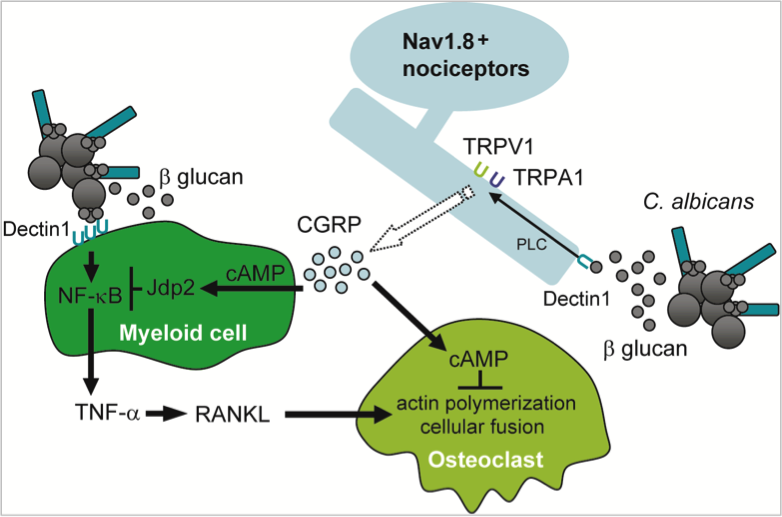
Pain neurons exaggerate inflammation of conditions like contact dermatitis and psoriasis. Despite its importance in allergic and autoimmune inflammation, it is not known so far whether pain neurons modulate pathogen-induced inflammation. Scientist at the Immunology Frontier Research Center (IFReC) discovered that the Nav1.8 ion channel-expressing pain neuron inhibits fungal inflammation and bone destruction.
"We generated the Nav1.8 ion channel-expressing neuron null mice. After C. albicans or C. albicans derived b-glucan injection into the hind paw, these mice showed significantly increased footpad swelling and bone destruction," said Kenta Maruyama, M.D., Ph.D. (Assistant Professor, IFReC).
Intriguingly, the Nav1.8 ion channel-positive neuron expresses Dectin-1, a b-glucan receptor, and Dectin-1 mediated inflammation is potently suppressed by pain neurons. Detailed experiments of pain neurons revealed that Dectin-1-stimulated pain neurons produce robust amounts of CGRP, a neuropeptide that inhibits osteoclast and cytokine production via TRPV1 and TPRA1 ion channel activation. "To our surprise, TRPV1/TRPA1 double deficient mice exhibited exaggerated inflammation and bone destruction in response to b-glucan due to impaired CGRP production, and myeloid cell transcription factor Jdp2 is necessary for the immunosuppression triggered by this neuropeptide," said Kenta.
A significant discovery in this study is that transcription factor Jdp2 is induced by CGRP and directly inhibits b-glucan-indeuced NF-kB activation in macrophages. Such findings are consistent with the results showing that Jdp2 deficiency hyper inflammatory phenotype induced by b-glucan was only observed in vivo.
Additional study revealed that β-glucan-induced CGRP production from pain neurons is more potent than that induced by LPS, a gram negative bacterial component.
"Previous reports suggested that pain neurons are deleterious for inflammation, but our findings suggest that pain neurons may function primarily in suppressing fungal inflammation, rather than bacterial inflammation" said Kenta. "Congenital insensitivity to pain with anhidrosis is an extremely rare hereditary disease characterized by impairment of nociceptor development. Manifestations of this disease are recurrent episodes of skin injury, osteomyelitis, bony fractures, and oral osteolysis. Our discovery may improve the therapeutic strategies of this disease and precise microbiological observation of this patient may clarify the bona-fide role of human pain neurons in fungal infection."
More information: Kenta Maruyama et al. Nociceptors Boost the Resolution of Fungal Osteoinflammation via the TRP Channel-CGRP-Jdp2 Axis, Cell Reports (2017). DOI: 10.1016/j.celrep.2017.06.002