Spreading depolarizations trigger early brain injury after subarachnoid hemorrhage, researchers find
The phrase "time is brain" could take on new meaning when applied to the treatment of subarachnoid hemorrhage, a type of bleeding stroke, thanks to research partially funded by the Mayfield Education & Research Foundation and the United States government.
Subarachnoid hemorrhage, caused by the rupture of a brain aneurysm, affects an estimated 10 to 15 of every 100,000 individuals each year. An international team of researchers has established that the blood from this type of hemorrhage launches deadly "brain tsunamis" within hours, leading to permanent brain damage. This damage, known as infarction, occurs in the cerebral cortex near the hemorrhage and can cause permanent disability and in some cases death.
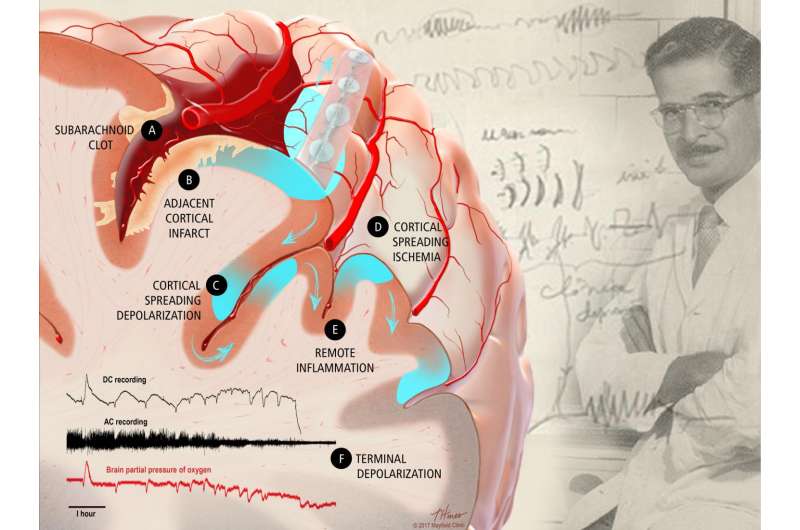
Brain tsunamis, scientifically known as cortical spreading depolarizations, are travelling waves of brain dysfunction that spread out from an injury site and contribute to worse outcomes in patients. They affect patients who suffer trauma to the brain as well as those who suffer various types of stroke. In subarachnoid hemorrhage, blood from a ruptured aneurysm or shredded artery pools in the space between the arachnoid membrane and the brain itself.
A team of researchers led by Principal Investigator Jed Hartings, PhD, Research Associate Professor in the Department of Neurosurgery at the University of Cincinnati, used a novel animal model to study what happens immediately after a subarachnoid hemorrhage. The team also monitored 23 patients who were surgically treated for subarachnoid hemorrhage at hospital locations of Charité University Medicine in Berlin. Monitoring was accomplished by placing electrode strips on the surface of the brains of animal models as well as patients.
The team's findings of secondary damage within 6 hours were published online today in the journal Brain.
"We found that patients who suffered brain damage in their frontal lobes were more likely to have experienced spreading depolarizations than those who had no damage," Dr. Hartings says. "The animal studies showed that these pathological changes can arise as a direct result of the blood accumulation in the grooves of the brain – that the presence of blood in the wrong place is toxic to the brain."
In the past, researchers have focused primarily on delayed complications—such as the narrowing of major cerebral arteries known as vasospasm— that occur 5 to 14 days after subarachnoid hemorrhage. Those complications account for only a minority of deaths (13 percent), however; the majority of deaths from subarachnoid hemorrhage (86 percent) occur in the early hours and days after an aneurysm rupture occurs. The present study sheds new light on these early events and suggests that they may be preventable.
"This is the first evidence that brain tsunamis are a clinical marker, and mechanism, of early brain injury," Dr. Hartings says. "As such, they provide us with an opportunity to pursue therapeutic interventions that could improve outcomes for patients. By treating or preventing brain tsunamis, we could potentially stop many victims of bleeding stroke from suffering additional, often catastrophic, brain damage."
The early damage caused by subarachnoid hemorrhage has been underappreciated, Hartings says, because it often cannot be observed with routine CT brain imaging. "There has also been a sense that early damage could not be prevented, but perhaps that perception is starting to change."
Looking ahead, the researchers propose the investigation in animal models of therapies that target spreading depolarizations in an effort to interrupt or prevent secondary injury processes.
When a brain injury occurs, nerve cells in the brain (which act like batteries by storing electrical and chemical energy) malfunction and effectively short-circuit. Because all nerve cells in the brain are connected, this depolarization causes all the neighboring cells to short-circuit as well; this subsequent leakage of precious electrical charge moves like a tsunami through the brain, with the potential to cause additional permanent tissue damage.
To document the cascade of events that immediately follows subarachnoid hemorrhage, the research team used a novel swine model, whose brain, with grooves and fissures, more closely resembles the human brain than previously studied rodent models. The researchers found that clots that quickly formed in the grooves of the swine brain mirrored the results in the human brain; in both situations, the clots caused repetitive spreading depolarizations and lesions (infarcts) in the cerebral cortex shortly after an aneurysm rupture and subarachnoid hemorrhage.
The study's collaborating scientists and clinicians are members of COSBID (Co-Operative Studies on Brain Injury Depolarizations.
More information: Ayata C, Lauritzen M. Spreading Depression, Spreading Depolarizations, and the CerebralVasculature. Physiological Reviews. 2015;95(3):953-93.
Baechli H, Behzad M, Schreckenberger M, Buchholz HG, Heimann A, Kempski O, et al. Blood constituents trigger brain swelling, tissue death, and reduction of glucose metabolism early after acute subdural hematoma in rats. J Cereb Blood Flow Metab. 2010;30(3):576-85.
Bosche B, Graf R, Ernestus RI, Dohmen C, Reithmeier T, Brinker G, et al. Recurrent spreading depolarizations after subarachnoid hemorrhage decreases oxygen availability in human cerebral cortex. Ann Neurol. 2010;67(5):607-17.
Bretz JS, von Dincklage F, Woitzik J, Winkler MKL, Major S, Dreier JP, et al. The Hijdra scale has a significant prognostic value for the functional outcome of Fisher grade 3 patients with subarachnoid hemorrhage. Clin Neuroradiol. 2016:in press
Hartings JA. Spreading depolarization monitoring in neurocritical care of acute brain injury. Curr Opin Crit Care. 2017;23(2):94-102.
Hartings JA, Shuttleworth CW, Kirov SA, Ayata C, Hinzman JM, Foreman B, et al. The
continuum of spreading depolarizations in acute cortical lesion development: Examining Leao's legacy. J Cereb Blood Flow Metab. 2017;37(5):1571-94.