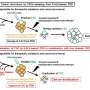
Areas of glioblastoma tumors correlate with separate subtypes of glioma stem cells
A new study published in the Oct. 9 issue of the journal Nature Medicine demonstrates, for the first time, that glioblastoma (GBM), the most common and most lethal brain tumor, is driven by two distinct subsets of cancer stem cells. Moreover, each subtype of glioma stem cells is driven by distinct transcriptional programs for growth and treatment resistance, and these different cell populations correspond to well-known morphological differences within the GBM itself.
More importantly, the researchers found that while chemotherapeutic agents targeting each subtype achieve modest efficacy alone, they are synergistic when combined as demonstrated in a mouse model.
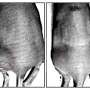
Senior co-author of the study, Andrew Sloan, MD, Medical Director, Brain Tumor and Neuro-Oncology Center at University Hospitals Cleveland Medical Center, observed that GBMs typically have two radiologically distinct regions on MRI: The enhancing mass and the necrotic core.
He noted, "We hypothesized that the different parts of the tumor might respond differently to treatment. Our findings are consistent with this hypothesis."
Even the best surgeons can only remove the entire tumor about 75 percent of the time, according to Dr. Sloan. Therefore, it is essential to learn more about the mechanisms for tumor growth and how the chemo agents can inhibit the pathways for growth.
Dr. Sloan said, "The enhancing edge and the invading tumor are driven by glioma stem cells (GSCs) dependent on the EZH2 pathway. They are driven by stem cells with the proneural pattern of gene expression and depend on rapid cell division and angiogenesis—creation of new blood vessels.
"Conversely, the necrotic part of the tumor is driven by a distinct set of glioma stem cells utilizing the BIM1 pathway and are characterized by a mesenchymal, inflammatory cell type dependent on glucose metabolism in the absence of oxygen.
"Inhibition of the EZH2 pathway slows growth of tumors in mouse brain derived from glioma stem cells from the enhancing margin of human tumors. Conversely, BIM1 inhibitors inhibit growth of mouse brain tumors derived from human glioma stem cells derived from the necrotic core of brain tumors in mice
Dr. Sloan said that since a single tumor may contain different pools of glioma stem cells, combined targeting should be considered for the pathways and stem cells.
The paper is entitled, "Targeting glioma stem cells through combined BMI1 and EZH2 inhibition."
Dr. Sloan is holds the Peter D. Cristal Chair in Neurosurgical Oncology and is Professor and Vice Chair, Department of Neurological Surgery at UH Cleveland Medical Center. He is also part of the Case Comprehensive Cancer Center, Case Western Reserve University School of Medicine.
Co-senior author is Jeremy Rich of the Division of Regenerative Medicine at the University of San Diego, and formerly at the Case Comprehensive Cancer Center. Lead author is Xun Jin of the Cleveland Clinic, Tianjin Medical University Cancer Institute and Hospital and the First Affiliated Hospital of Wenzhou Medical University, P.R. China.
More information: Targeting glioma stem cells through combined BMI1 and EZH2 inhibition, Nature Medicine (2017). nature.com/articles/doi:10.1038/nm.4415