Study finds link between fragile X syndrome gene and dysregulated tissue growth
Researchers at Indiana University have found a previously undetected link between the gene that causes fragile X syndrome and increased tissue growth. The link could reveal a key biological mechanism behind the serious physical and mental impairments caused by the disorder.
An inheritable genetic condition, fragile X syndrome is estimated to cause mild to moderate intellectual disabilities in 1 in 4,000 to 5,000 males and 1 in 6,000 to 8,000 females. It also causes physical abnormalities such as large brain size and weight at birth, unusually fast growth in height, gastrointestinal issues and high risk of obesity.
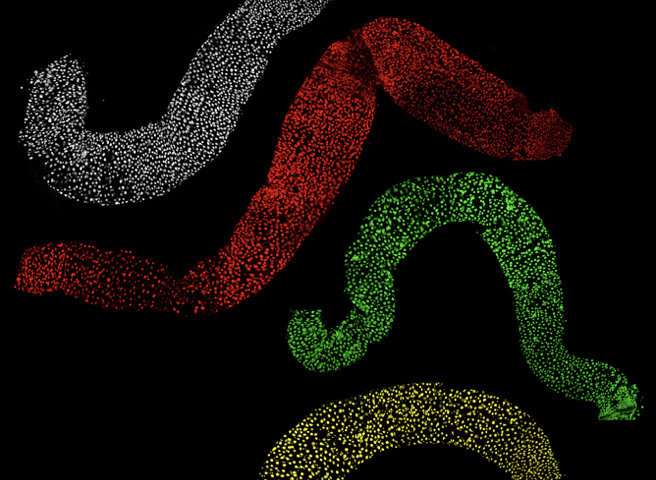
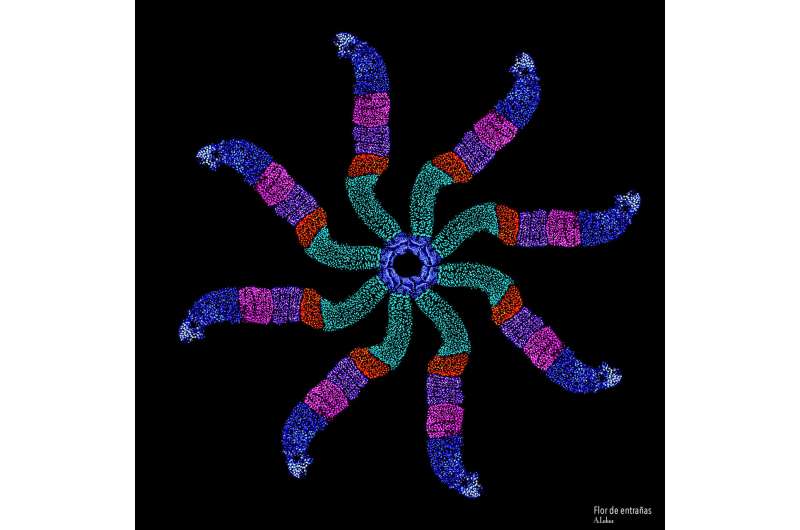
The study, which was conducted in fruit flies, focused on the effects of losing the same protein that's missing in people with fragile X syndrome on the intestines of insects modified to model the disease. The work is reported in the journal Cell Reports.
"To our knowledge, this is the first study to find a stem-cell-based mechanism by which a protein that is absent in people with fragile X syndrome limits excessive organ growth," said Arthur Luhur, a research associate in the IU Bloomington College of Arts and Sciences' Department of Biology, who is the lead author on the study. "This could represent a root cause for the gastrointestinal problems seen in people with the condition."
Similarly, Luhur said the stem-cell-based mechanism could cause symptoms seen in other parts of the body in fragile X syndrome, such as accelerated height growth from unrestrained activity in the bones' stem cells, or mental problems caused by excess neurons.
He also noted that basic biological research into fragile X syndrome's non-neurological symptoms is important since these effects are understudied—and therefore undertreated—compared to problems with mental development.
Fragile X syndrome is caused by an error in a single gene in the X chromosome called FMR1. Typically, the FMR1 gene creates a protein called FMRP. In individuals with fragile X syndrome, an abnormality in this gene causes the body to "silence" the production of FMRP.
The IU team found that the absence of FMRP causes a higher rate of cellular growth—driven by symmetric stem cell division. Typically, this process is balanced by asymmetric stem cell division, when old cells are replaced with new ones.
"The cells that lacked FMRP acted like cars without brakes," said Nicholas Sokol, an associate professor in the Department of Biology, who is the senior author on the study. "They were ready to divide more often and more quickly, and they tended to divide symmetrically, causing the intestine to be bigger than normal."
The discovery grew from the researchers' earlier research into another gene, called LIN-28, that also appeared to affect growth in the intestinal cells in fruit flies. During that study, IU researchers came across evidence that FMRP—the protein encoded by the gene that causes fragile X syndrome—played a role in the same biomolecular pathway as LIN-28.
The earlier study found that lower levels of LIN-28 reduced insulin receptors in the intestines. The reduced number of insulin receptors slowed cellular growth. The new study found that stem cells from the intestines of flies whose genes contained the genetic mutation for fragile X syndrome activated these insulin receptors at a higher rate than normal cells. Moreover, the IU scientists found that higher levels of FMRP affected LIN-28 levels, but not vice versa, suggesting that FMRP plays a controlling role in the biological pathway.
The research may also open doors to research on new treatments of fragile X syndrome, Luhur said. For example, the study found that normal cell growth could be restored by reducing the insulin-signaling activity in fruit flies' intestines using genetic interventions or changes in nutrition. The connection between insulin receptor activity and intestinal growth may also provide new insight into other research, which has shown that the FDA-approved diabetes drug Metformin seems to alleviate some of the neurological symptoms of fragile X syndrome.
"The next step is conducting additional research on FMR1 and LIN-28 in animals to learn more about their biochemical relationship and their effect on metabolism in the body," Luhur said. "These are important questions for understanding how their interaction affects physiology and human health."
More information: Cell Reports (2017). DOI: 10.1016/j.celrep.2017.11.039 , www.cell.com/cell-reports/full … 2211-1247(17)31676-5