February 8, 2018 report
A possible way to protect intestinal walls from damage due to chemotherapy
A team of researchers at the University of Pittsburgh has found a possible way to protect cells in the intestines when subjected to chemotherapy. In their paper published in Science Translational Medicine, the team describes their approach, how well it worked in mice, and obstacles yet to be overcome.
Chemotherapy is the administration of a concoction of drugs to kill cancerous tumors. But it also has some negative side effects such as hair loss and vomiting. In this new effort, the researchers have been looking into ways to reduce the damage to intestinal cells walls from chemotherapy, both because it would make treatment more bearable and because it would allow for higher doses of chemotherapy.
The intestines are lined with cells that protect against stomach acid and bacteria, and because of that, must be replaced more often than other cells—that means that the intestines have a large supply of stem cells. Unfortunately, chemotherapy causes DNA damage to intestinal wall cells, setting off p53, a tumor suppressor. Once p53 is activated, signals induce the creation of the proteins PUMA and p21, which in turn signal the stem cells to begin apoptosis (cell suicide). Prior research has shown that preventing p53 from activating is not a viable option because it would shield tumors from the chemo drugs meant to kill them. In this new effort, the researchers looked instead for a way to prevent PUMA from being created, thus preventing the stem cells from killing themselves off.
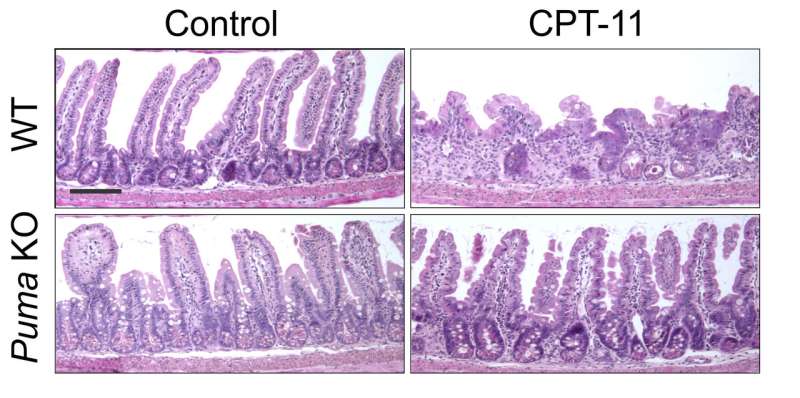
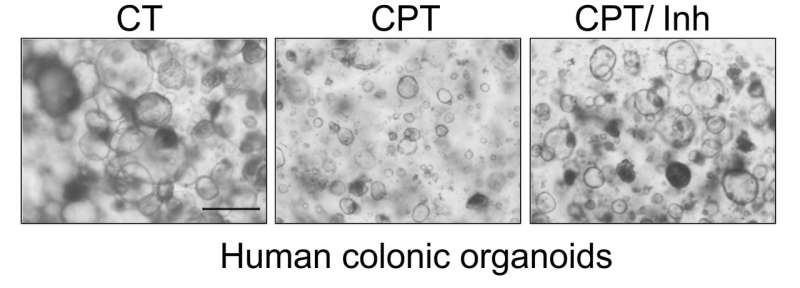
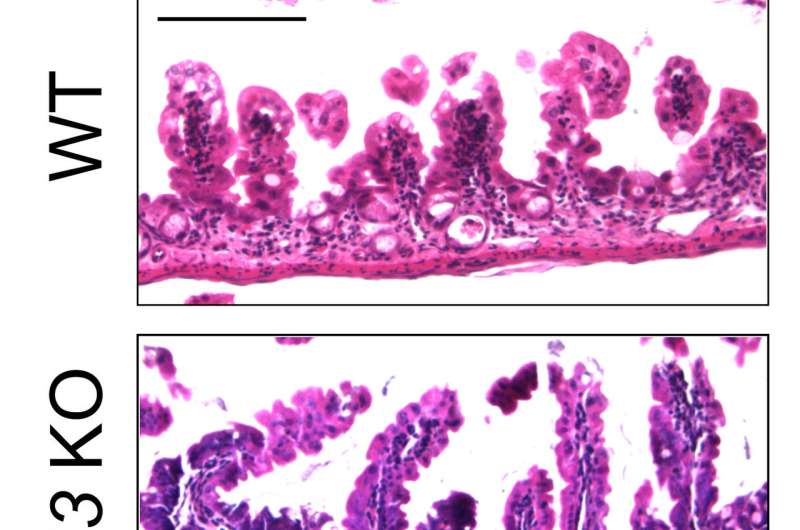
To test this approach, the team engineered PUMA protein-knockout mice and then gave them chemotherapy to see how their intestinal cells fared. The researchers report that there was far less damage than there would have been under normal conditions. Next, the team tested a small molecule that prior research had suggested inhibits the production of PUMA by giving it to healthy normal test mice, then followed up with chemo drugs. They report that once again, the mice showed far less damage to intestinal stem cells. The final test consisted of inhibiting the protein in cultured colon cells in a petri dish and then dousing them with chemo drugs—once again, the approach appeared to work as hoped.
Emboldened by their results, the team plans to continue testing the approach to find out if it might turn out to be a viable option for human cancer patients.
More information: Brian J. Leibowitz et al. Targeting p53-dependent stem cell loss for intestinal chemoprotection, Science Translational Medicine (2018). DOI: 10.1126/scitranslmed.aam7610
Abstract
The gastrointestinal (GI) epithelium is the fastest renewing adult tissue and is maintained by tissue-specific stem cells. Treatment-induced GI side effects are a major dose-limiting factor for chemotherapy and abdominal radiotherapy and can decrease the quality of life in cancer patients and survivors. p53 is a key regulator of the DNA damage response, and its activation results in stimulus- and cell type–specific outcomes via distinct effectors. We demonstrate that p53-dependent PUMA induction mediates chemotherapy-induced intestinal injury in mice. Genetic ablation of Puma, but not of p53, protects against chemotherapy-induced lethal GI injury. Blocking chemotherapy-induced loss of LGR5+ stem cells by Puma KO or a small-molecule PUMA inhibitor (PUMAi) prevents perturbation of the stem cell niche, rapid activation of WNT and NOTCH signaling, and stem cell exhaustion during repeated exposures. PUMAi also protects human and mouse colonic organoids against chemotherapy-induced apoptosis and damage but does not protect cancer cells in vitro or in vivo. Therefore, targeting PUMA is a promising strategy for normal intestinal chemoprotection because it selectively blocks p53-dependent stem cell loss but leaves p53-dependent protective effects intact.
© 2018 Phys.org