Muscle regeneration compromises stability in muscular dystrophy
A new study finds that muscle fibers in Duchenne muscular dystrophy (DMD) split during regeneration to such an extreme that the muscle is weakened beyond repair. The article is published ahead of print in the American Journal of Physiology—Cell Physiology.
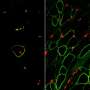
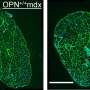
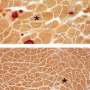
DMD is a genetic degenerative disorder that affects mostly boys. People with DMD do not make dystrophin, a protein that helps keep the muscles intact. Without dystrophin, muscles constantly break down and regenerate. Muscle regeneration, even in healthy people, causes some of the fibers to split into multiple strands called branches. Branching typically helps the new muscle become bigger, stiffer and overall stronger. Researchers from Australia found that this may not be the case in DMD.
The research team studied muscle fibers in two mouse models of DMD. One group of mice was younger than the other. The researchers found extensive branching in the older group. Some muscle fibers in DMD can have as many as 10 separate branches, explained Stewart Head, Ph.D., one of the study's authors. Muscle contraction tests caused the branched fibers to rupture, or tear apart, in the older mice. The younger mice had fewer and less complex branched fibers and did not experience rupturing, showing that "a moderate amount of simple branching does not compromise the mechanical stability of the muscle," Head said.
"This evidence supports our concept of 'tipping point' where the number and extent of fiber branching reaches a level where the branching itself terminally compromises muscle function, irrespective of the absence of dystrophin," the researchers wrote.
The full article, "Branched fibers from old fast-twitch dystrophic muscles are the sites of terminal damage in muscular dystrophy," is published in the American Journal of Physiology—Cell Physiology.
More information: Leonit Kiriaev et al. BRANCHED FIBERS FROM OLD FAST-TWITCH DYSTROPHIC MUSCLES ARE THE SITES OF TERMINAL DAMAGE IN MUSCULAR DYSTROPHY, American Journal of Physiology-Cell Physiology (2018). DOI: 10.1152/ajpcell.00161.2017