RNA-based therapeutic inhibits a metabolic pathway in tumor-initiating lung cancer cells
RNA-based therapeutics that prevent a key metabolic enzyme from being expressed in tumor-initiating cells (TIC) hold promise for the treatment of lung cancer, an A*STAR team has shown.
As implied by their name, TIC – also known as cancer stem cells – are a subset of tumor cells with the capacity to self-renew and start new tumors. Abnormal metabolism is a key feature of TIC as the high proliferation rate and tumorigenic potential of these cells require changes to the metabolic pathways that are normally used by healthy cells to produce energy and biosynthetic precursors.
The discovery of elevated expression of normal or mutant forms of metabolic enzymes in a variety of cancers has created great interest in cancer metabolism, explains Uttam Surana from the Institute of Molecular and Cell Biology, and Dave Wee from the Institute of High Performance Computing. Targeting TIC metabolism is emerging as a promising strategy to thwart the progression of various cancers.
Previous studies have shown that TIC of non-small cell lung carcinoma (NSCLC) tumors contain high levels of glycine decarboxylase (GLDC), an enzyme that breaks down the amino-acid glycine. Overexpression of GLDC stimulates the generation of tumors and the proliferation of TIC, and high GLDC levels are associated with poor survival rates in NSCLC patients.
No therapeutic agents have been developed against GLDC until now. Surana and Wee identified short synthetic RNA sequences (steric hindrance antisense oligonucleotides, or shAONs) that supressed the production GLDC protein in human lung cancer cells, hindering their proliferation and preventing tumor formation.
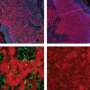
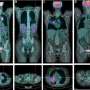
Working with colleagues at A*STAR's Genome Institute of Singapore, the team showed that when the GLDC-targeting shAON sequences were injected into mice bearing NSCLC enriched with TIC, tumor growth was reduced by 60 percent compared with mice injected with scrambled control sequences.
The shAONs disrupt a key step in the process through which the gene that encodes GLDC is translated into protein. "We have designed three shAONs that efficiently interfere with the process that normally removes non-protein coding sequences from the GLDC pre-mRNA transcribed from the gene's DNA template," Surana explains. "The resultant aberrant transcripts are primed for degradation and GLDC protein production is dramatically reduced".
Future work will determine whether these shAONs can be delivered through the nose to target tumors grafted in the lungs of mice, and test their efficacy against other types of cancer in which an upregulation of GLDC contributes to TIC metabolism, such as breast and thyroid cancers. Because of shAONs' high target specificity, they offer important advantages over small molecule drugs, which have a higher risk of resistance and toxicity.
More information: Jing Lin et al. Induced-Decay of Glycine Decarboxylase Transcripts as an Anticancer Therapeutic Strategy for Non-Small-Cell Lung Carcinoma, Molecular Therapy - Nucleic Acids (2017). DOI: 10.1016/j.omtn.2017.10.001