Stem cell reserves found in the stomach have implications for the genesis of gastric cancer
A team of researchers at A*STAR has found that certain cells in the stomach, called chief cells, become stem cells in response to injury, providing a source of new cells. As well as being significant for understanding how the stomach lining renews itself, the finding sheds light on how gastric cancer can begin.
The stomach wall is a hostile place for cells, as they are constantly subjected to mechanical stress and high acidity and must be continually replaced. The most likely source of such reinforcements is adult stem cells, but identifying these cells has been challenging.
"The identity of the gastric stem cells has been contentious," says Nick Barker of the A*STAR Institute of Medical Biology.
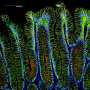
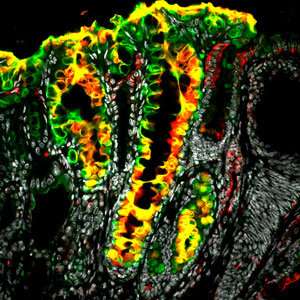
Now, Barker and his co-workers have located where these stem cells reside—at the bottom of small pits in the stomach known as corpus glands. They discovered in a mouse model that certain cells in this location express the gene Lgr5, a marker of stem cells in various organs, including the intestine and skin. The researchers also found that these cells are not normally stem cells; rather, they become stem cells in response to damage to the stomach wall.
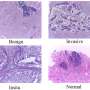
In addition, the team has implicated these cells in the genesis of gastric cancer—the fourth leading cause of cancer globally, according to the World Health Organization. They discovered that when these cells undergo a mutation, they become a major source of tumors at the base of the corpus glands (see image).
"The theory that chief cells can be activated to serve as the precursor to gastric cancer under damage conditions is controversial," says Barker. "Indeed, a recent study concluded that other cells are the true precursors of cancer, not chief cells. In this study, we conclusively identified Lgr5+ cells as a source of spasmolytic polypeptide-expressing metaplasia (SPEM), helping to clarify the ongoing debate."
In the future, the team intends to explore exactly what causes the cells to become stem cells. "We are interested in better understanding the mechanism responsible for converting the normally quiescent, differentiated chief cells into proliferative, multipotent stem cells," says Barker. He notes that the same mechanism could be at play when the cells become cancerous. "We hope that these findings will contribute to translatable clinical outcomes," he adds.
More information: Marc Leushacke et al. Lgr5-expressing chief cells drive epithelial regeneration and cancer in the oxyntic stomach, Nature Cell Biology (2017). DOI: 10.1038/ncb3541