Team develops 3-D tissue model of a developing human heart

The heart is the first organ to develop in the womb and the first cause of concern for many parents.
For expecting mothers, the excitement of pregnancy is often offset by anxiety over medication they require. Parents and doctors often have to consider the mother's health as well as the potential risk regarding how medication could affect their baby. The U.S. Food and Drug Administration requires certain drugs to be labeled with pregnancy exposure and risk. Some drugs are labeled to show that testing on animals has failed to demonstrate a risk but there are no adequate and well-controlled studies of pregnant women.
"Some drugs are difficult for doctors to prescribe to pregnant women because they don't know the embryo toxicity, how does that effect fetal development," said biomedical engineering Professor Zhen Ma. "They don't have the clinical outcome based on human study."
Ma and his research team in Syracuse University's System Tissue Engineering & Morphogenesis (STEM) lab have been working with human induced pluripotent stem cells to study tissue regeneration, regenerative medicine and stem cell engineering.
"This type of stem cell has the ability to generate all the different cells in a human body. Because it was derived from humans," said Ma.
Pluripotent cells can be used to create heart tissue but Ma's research team believed they could take it even further.
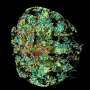
"We can try to rebuild the shape of the early development heart in the lab," said Ma. "It mimics the very early stage, during the embryo genesis - how the heart was formed."
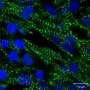
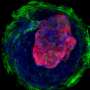
Ma's research team developed a process that combines biomaterials-based cell patterning and stem cell technology to make a 3D tissue model that could mimic early stage human heart development. By starting with a layer of polymer in a tissue culture dish and etching tiny patterns in the polymer, the stem cells will only attach within those patterns. Since the stem cells do not attach to the polymer, they grow within the patterns and eventually develop into a three-dimensional structure that has distinct tissue types. The process developed by Ma's team focused on cardiac tissue but other labs could adapt it to other tissue types and even organ tissues.
Their research will be published in the March 2018 Nature Protocols journal and featured on the cover.
The platform allows tissue to form during the cell differentiation process rather than building tissue out of already established heart cells. Tissue that forms during the differentiation process has more layers and more accurately represents how tissue naturally develops in humans.
"Using the cell lines we use, they are human based so we know they will affect human tissue in a certain way as opposed to the uncertainty that comes with an animal model," said graduate student Plansky Hoang.
Some pregnant women avoid taking drugs they need to manage chronic conditions but if the mother's health suffers, that can also affect her baby. More reliable test results could provide more confidence for both patients and doctors.
"It helps people make better decisions," said Ma. "If we can determine it is safe, it should be prescribed to women who need these drugs."
Embryotoxicity is just one potential use of the modeling platform developed by Ma and his team. Countless other human tissues could also be cultured using the process. It could also allow for individualized drug toxicity testing for humans. Different people can have different reactions to the same drug but personalized testing using someone's stem cells could help determine if a drug is safe for them before they take it.
"The traditional way of screening, they take a patient history and then test you on a drug for a month or two and they assess again you after that," said Hoang. "By using our model we can test for multiple drugs at once so if there is a series of drugs that will potentially benefit you, we can test all of them at once as opposed to one at a time that takes longer."
More information: Plansky Hoang et al. Generation of spatial-patterned early-developing cardiac organoids using human pluripotent stem cells, Nature Protocols (2018). DOI: 10.1038/nprot.2018.006