Using mathematical models to determine the best chemotherapy schedules

Can mathematical models predict how can cancer cells respond to varied chemotherapy schedules? In other words, should cancers associated with fast-growing tumors, like brain cancer, be treated using a low drug dose administered continuously, as opposed to a high drug dose given periodically?
Professor Paul Newton and Ph.D. recipient Jeffrey West have developed a tool that can predict the best course of treatment for cancer patients. The method goes beyond what previous mathematical models were capable of by applying evolutionary information and using cell-based data. And because the researchers can run computer simulations based on their model, they aren't subject to the restrictions associated with clinical trials.
In their latest study, published in the journal Cancer Research, Newton and West determined that chemotherapeutic schedules should be designed based on the growth rate of the tumor.
"What we're doing is trying to design chemotherapeutic schedules to better overcome some of the known problems associated with chemotherapy," said Newton, professor of aerospace and mechanical engineering, mathematics and medicine—the latter at the Norris Comprehensive Cancer Center at the Keck School of Medicine of USC. He also teaches at the USC Dornsife College of Letters, Arts and Sciences.
Chemotherapy schedules and resistance
One of these problems, which has become a focus of their research, is chemotherapeutic resistance by competitive release.
Traditionally, to reduce and potentially eliminate tumors, patients are given a chemotherapy drug and a schedule that targets all cancer cells. They receive this drug until the tumor is gone, most commonly through a high dose once every two or three weeks—also known as a maximum tolerated dose, or MTD, schedule—but sometimes through a low-dose, continuous treatment plan known as low-dose metronomic, or LDM.
However, what often occurs during these treatment plans is an initial shrinking of the tumor, indicating that they are seemingly on the correct path, followed by tumor regrowth despite the patients are receiving. The cause of this recurrence is due to the composition of the tumor itself.
"It's not just one kind of cancer cell in your tumor," Newton said. "It's a whole bunch of different kinds of cancer cells that are all competing at different growth rates."
Cells can divide at will
After the chemotherapy drug has eliminated enough of the most prolific cancer cell types, the other cells that have survived are resistant to the drug and now have less competition for space and nutrients. These resistant cells have been "released from competition" and are now free to grow and divide at will. The next round of chemotherapy will therefore be far less effective.
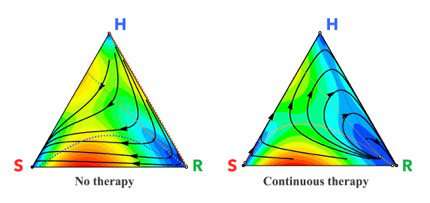
"When you go in and take a biopsy or do imaging of the patient's tumor, you can't always count the number of resistant cells or tell which of those cells are going to be resistant," West said. "One of the points of making a mathematical model is to predict things that can't be directly monitored in the clinic."
Their predictive tool allows them to test different chemotherapy plans on a tumor while accounting for the changing cell populations.
To do so, they used existing data on cancers treated by either MTD or LDM and modeled the competition between cells using evolutionary game theory, a mathematical framework that uses Charles Darwin's concept of "survival of the fittest." The model picks two cells at random for a head-to-head competition in which the winner is determined based on "fitness," which is dependent on the cell type's growth rate.
The players are the healthy cells, the targeted cancer cells and the resistant cancer cells and the game is played over and over again in the computer simulation as the different populations of cells compete. For each treatment scenario, the model hosts millions of competitions to simulate how the tumor will most likely progress over time.
"We cycled through all possible combinations of scheduling and different drug concentrations, and we sort of created a histogram of what would be the most likely benefits in different scenarios," Newton said.
Fast-growing tumors
The duo found that chemotherapeutic schedules should be determined by the growth rate of the tumor, something not currently done in a clinical setting. For example, fast-growing tumors, typical with brain cancer, are better controlled using LDM, while MTD worked better against slow-growing tumors, typical with prostate cancer.
In addition to comparing existing therapies, the researchers will use their model to develop treatment plans that adapt as the tumor changes. This requires monitoring the different cell populations that make up the growing tumor and adjusting the schedule based on the data. In engineering circles, this approach is called "closed-loop" control.
"The hope for the future is to have patient-specific models where a patient walks into a clinic and you can type in their information and then run the model … and predict the optimal schedule and drugs," West said.
After completing his Ph.D. in mechanical engineering in August, West is now working as a postdoctoral researcher for the Moffitt Cancer Center in Florida. Working in the Integrative mathematical oncology department, he is using the mathematical model to test treatment plans being developed by clinicians at the center before they are applied to patients during clinical trials.
"The way that new clinical trials are designed is in large part based on the intuition of the clinician, but the timelines of these things can be years to even decades," West said. "Math models can sort of close the loop and test the clinician's intuition in mathematical abstraction before the clinical trial even starts."
More information: Jeffrey West et al. Chemotherapeutic Dose Scheduling Based on Tumor Growth Rates Provides a Case for Low-Dose Metronomic High-Entropy Therapies, Cancer Research (2017). DOI: 10.1158/0008-5472.CAN-17-1120

















