Researchers reveal why Hippo pathway may have an outsized effect on solid tumor

Like detectives tracking a serial killer, St. Jude Children's Research Hospital scientists have identified a novel mechanism that may help promote the growth and spread of solid tumors. The findings appeared online Oct. 25 in the journal Developmental Cell.
The research focused on the transcription factors YAP and TAZ. The proteins, which have overlapping functions, activate gene expression and drive cell proliferation. YAP/TAZ are controlled by the Hippo signaling pathway. The pathway is altered in many solid tumors, leading to activation of YAP/TAZ.
But how YAP/TAZ drives proliferation and contributes to tumor growth and spread have been unclear.
Xinwei Cao, Ph.D., an associate member of the St. Jude Department of Developmental Neurobiology, and her colleagues have a possible answer: hypertranscription.
"Rather than activating a few genes involved in cell proliferation, activation of YAP/TAZ in this study caused a global increase in gene transcription activity, or hypertranscription," said Cao, the paper's corresponding author. "Overall cellular transcription activity increased by about 50 percent.
"Many of the genes activated are involved in biosynthesis and make the proteins, RNA and other materials necessary for cell growth and proliferation."
While hypertranscrption has been reported in cancer cells, this study suggests for the first time the process might explain how inactivation of the Hippo pathway fuels tumor growth. "The hypertranscription phenomenon was known, but had not previously been associated with YAP/TAZ," Cao said.
The process
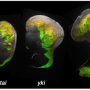
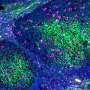
For this study, researchers activated YAP/TAZ in immature brain cells (neural progenitor cells) in mice by deleting two genes in the Hippo pathway. The genes, Lats 1/2, are the last line of defense in the pathway to prevent activation of YAP/TAZ.
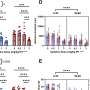
Scientists used a method called cell-number-normalized transcriptome analysis to compare how activation of YAP/TAZ affected gene expression levels in normal and mutant mouse neural progenitor cells. The method accounts for differing amounts of total RNA in the mutant and normal cells. A conventional analytic tool failed to detect the global changes in transcription and the genes involved.
A surprise
Prior research in other organs found that disruption of the Hippo pathway and activation of YAP/TAZ led to cell proliferation and tumor formation.
"To our surprise, the complete opposite occurred in neural progenitor cells," Cao said. "There was a transient increase in cell proliferation, followed by massive apoptosis in the developing brain."
Scientists took a closer look and found that in neural progenitor cells, hypertranscription triggered replication stress, DNA damage and cell death. The cell death was caused by apoptosis, the programmed cell death pathway that helps the body get rid of unneeded, unwanted or dangerous cells.
"Neural progenitor cells may be uniquely sensitive to hypertranscription and have a mechanism to safeguard against over-proliferation, while the liver or other organs might be more likely to respond with tumor formation," Cao said.
Researchers are using these results to explore how YAP/TAZ activation may influence development of other solid tumors.
More information: Alfonso Lavado et al. The Hippo Pathway Prevents YAP/TAZ-Driven Hypertranscription and Controls Neural Progenitor Number, Developmental Cell (2018). DOI: 10.1016/j.devcel.2018.09.021