New compound could enhance the efficacy of standard breast cancer treatment

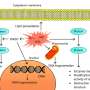
A novel compound against breast cancer improves the efficacy of standard cancer treatment and shows superior tolerability in laboratory tests when compared to similar agents, setting the stage for its further clinical development. The mechanism of action of this compound is based upon the re-activation of naturally occurring "programmed" cell death or apoptosis, that is switched off by many cancer cells thereby leading to their uncontrolled proliferation. Results of this study have now been published by physician-scientists from the Karl Landsteiner University of Health Sciences, Krems together with colleagues from the University of Heidelberg, Harvard Medical School, St. Jude's Children's Research Hospital, the Huazhong University and others.
For the development of novel cancer therapeutics it is essential to balance the pros (elimination of malignant cells) and cons (irreparable damage to healthy tissues). Findings now published by a research group led by Dr. Klaus Podar at the Karl Landsteiner University of Health Sciences, Krems are very likely to attract marked attention within the cancer research community. The group has demonstrated high anti-breast cancer activity and excellent tolerance of a novel, targeting agent, especially when used in combination with established cancer drugs. These findings build on Dr. Podar's and other groups' previous studies that have defined Mcl-1, an anti-apoptotic protein often overexpressed in tumor cells, to be a promising novel therapeutic target in breast cancer. The recent published data pave now the way for the further development of a derived novel agent for clinical application.
Therapy to induce cancer cell death
Apoptosis is a vital molecular mechanism to physiologically eliminate cells, thereby enabling normal cell turnover, proper development and functioning of the immune system, and chemical/ radiation- induced cell death. The active novel agent, called EU-5346, inhibits the binding of the anti-apoptotic protein Mcl-1 to the pro-apoptotic protein Bim. The binding of Mcl-1 to Bim suspends Bim-induced apoptosis. "The substance that we have tested makes breast cancer cells 'mortal' again," explains Dr. Sonia Vallet, the study's lead author from the Department of Internal Medicine II at the University Hospital, Krems "It therefore not only shortens the life of breast cancer cells but also prevents the development of resistance to certain cancer drugs; both positive effects when it comes to treatment."
Numerous research groups and pharmaceutical companies are currently focusing on the development of Mcl-1- targeting agents. However, their general clinical application has so far been hampered due to significant side effects, thrombocytopenia and cardiotoxicity in particular. Dr. Vallet and collaborators from the University of Heidelberg, Harvard Medical School, St. Jude's Children's Research Hospital, Huazhong University and others therefore also tested in vitro EU-5346- associated toxicity. Dr. Podar was pleased with the outcome: "We were all delighted when our results indicated that EU-5346 was comparatively less toxic to the heart, blood and nerves."
Tackling resistance
Cells often develop resistance to common drugs, including tamoxifen, trastuzumab and paclitaxel. Therefore, the group also tested the anti-tumor effects of EU-5346- containing drug combinations. Of note, results demonstrate that EU-5346- containing combinations with these drugs improve or even re-activate their anti-tumor effects. "Our findings show that EU-5346 is a promising candidate for further drug development in a clinical setting," said Dr. Vallet, summarizing the encouraging results of the international collaboration. "In addition to breast cancer, I am convinced that Mcl-1 inhibitors have also a high therapeutic potential other malignancies" Dr. Podar added. However, additional research is required to define the prevalence of Mcl-1 and other anti-apoptotic proteins in individual patients. These data will provide the rationale for personalized Mcl-1 inhibitor- containing combination therapies.
More information: Sonia Vallet et al. Rationally derived drug combinations with the novel Mcl-1 inhibitor EU-5346 in breast cancer, Breast Cancer Research and Treatment (2018). DOI: 10.1007/s10549-018-5022-5