Scientists discover 'brain hot spot' for drugs against fear
Anxiety disorders are a major medical problem affecting a large population. They can be treated with a range of psychiatric drugs, including a group of substances called benzodiazepines (BZDs). BZDs have been used to treat patients with anxiety for 50 years and it is well-understood what they do on the molecular and cellular level. However, doctors and neuroscientists had much less understanding of the neural circuit interactions through which BZDs unfold their anxiety relieving effect.
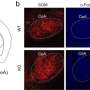
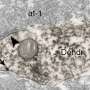
A team of scientists around Wulf Haubensak of the Research Institute of Molecular Pathology (IMP) in Vienna and Andreas Hess of the Friedrich-Alexander-Universität Erlangen-Nürnberg has now used a combination of innovative methods, linking genetics, information on neuronal circuits and functional brain mapping. They found that BZDs interfere with the relay of aversive signals through the amygdala and characterised the circuits involved. The study was now published in the journal Molecular Psychiatry.
"Fear emerges from the interaction of several circuits across the brain. In this network, we identified a crucial biomedical 'hot-spot' underlying fear-relieving therapy", said Wulf Haubensak. "Tracking down this hot-spot has only become possible by combining insights on the connections of neurons in the brain, the connectome, with genetic techniques that allow the functional visualisation and manipulation of specific populations of neurons in living animals – methods and information that have become available only very recently."
The scientists used mice for their experiments, but by comparing their findings to the functional brain scans of humans, they found clues that the same mechanisms are conserved in people. This opens new windows for drug development.
Andreas Hess of the Friedrich Alexander University and co-author of the study, emphasised the importance of functional brain imaging: "Non-invasive imaging techniques such as magnetic resonance imaging are key to study neurobiological functions at the whole brain level. We combined this with novel data analysis strategies to characterise the modulatory impact of small neuronal circuits that underly important brain function—in this case anxiety."
"Knowing the exact networks of neurons that mediate the BZD anxiolytic effect, we can now try to target them specifically. This may allow the development of new drugs that treat anxiety without the side effects common to current anxiolytics", says Johannes Griessner, Ph.D. candidate and first author of the study. He concludes with a broader outlook on how the findings could be used in further studies: "Psychiatry needs a strong biological basis which allows for targeted therapeutic interventions. Our approach could serve as a blueprint for an experimental strategy that could be used to better characterise the effects of psychoactive drugs in general."
More information: Johannes Griessner et al. Central amygdala circuit dynamics underlying the benzodiazepine anxiolytic effect, Molecular Psychiatry (2018). DOI: 10.1038/s41380-018-0310-3