Alzheimer's disease: It may be possible to restore memory function, preclinical study finds

Research published today in the journal Brain reveals a new approach to Alzheimer's disease (AD) that may eventually make it possible to reverse memory loss, a hallmark of the disease in its late stages.
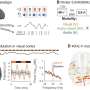
The team, led by University at Buffalo scientists, found that by focusing on gene changes caused by influences other than DNA sequences—called epigenetics—it was possible to reverse memory decline in an animal model of AD.
"In this paper, we have not only identified the epigenetic factors that contribute to the memory loss, we also found ways to temporarily reverse them in an animal model of AD," said senior author Zhen Yan, Ph.D., a SUNY Distinguished Professor in the Department of Physiology and Biophysics in the Jacobs School of Medicine and Biomedical Sciences at UB.
The research was conducted on mouse models carrying gene mutations for familial AD—where more than one member of a family has the disease—and on post-mortem brain tissues from AD patients.
AD is linked to epigenetic abnormality
AD results from both genetic and environmental risk factors, such as aging, which combine to result in epigenetic changes, leading to gene expression changes, but little is known about how that occurs.
The epigenetic changes in AD happen primarily in the later stages, when patients are unable to retain recently learned information and exhibit the most dramatic cognitive decline, Yan said. A key reason for the cognitive decline is the loss of glutamate receptors, which are critical to learning and short-term memory.
"We found that in Alzheimer's disease, many subunits of glutamate receptors in the frontal cortex are downregulated, disrupting the excitatory signals, which impairs working memory," Yan said.
The researchers found that the loss of glutamate receptors is the result of an epigenetic process known as repressive histone modification, which is elevated in AD. They saw this both in the animal models they studied and in post-mortem tissue of AD patients.
Yan explained that histone modifiers change the structure of chromatin, which controls how genetic material gains access to a cell's transcriptional machinery.
"This AD-linked abnormal histone modification is what represses gene expression, diminishing glutamate receptors, which leads to loss of synaptic function and memory deficits," Yan said.
Potential drug targets
Understanding that process has revealed potential drug targets, she said, since repressive histone modification is controlled or catalyzed by enzymes.
"Our study not only reveals the correlation between epigenetic changes and AD, we also found we can correct the cognitive dysfunction by targeting the epigenetic enzymes to restore glutamate receptors," Yan said.
The AD animals were injected three times with compounds designed to inhibit the enzyme that controls repressive histone modification.
"When we gave the AD animals this enzyme inhibitor, we saw the rescue of cognitive function confirmed through evaluations of recognition memory, spatial memory and working memory. We were quite surprised to see such dramatic cognitive improvement," Yan said.
"At the same time, we saw the recovery of glutamate receptor expression and function in the frontal cortex."
The improvements lasted for one week; future studies will focus on developing compounds that penetrate the brain more effectively and are thus longer-lasting.
Epigenetic advantage
Brain disorders, such as AD, are often polygenetic diseases, Yan explained, where many genes are involved and each gene has a modest impact. An epigenetic approach is advantageous, she said, because epigenetic processes control not just one gene but many genes.
"An epigenetic approach can correct a network of genes, which will collectively restore cells to their normal state and restore the complex brain function," she explained.
"We have provided evidence showing that abnormal epigenetic regulation of glutamate receptor expression and function did contribute to cognitive decline in Alzheimer's disease," Yan concluded. "If many of the dysregulated genes in AD are normalized by targeting specific epigenetic enzymes, it will be possible to restore cognitive function and behavior."
More information: Brain (2019). academic.oup.com/brain/article … 10.1093/brain/awy354