Study overturns dogma of cancer metabolism theory
Scientists at the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA have discovered that squamous cell skin cancers do not require increased glucose to power their development and growth, contrary to a long-held belief about cancer metabolism.
The findings could bring about a better understanding of many cancers' metabolic needs and lead to the development of more effective therapies for squamous cell skin cancer and other forms of epithelial cancer.
The research, led by senior authors Heather Christofk and Bill Lowry, was published in the journal Nature Communications.
A fundamental doctrine of cancer metabolism theory is that cancer cells are glycolytic, meaning they consume more glucose and produce more lactate than normal cells. This metabolic shift, called aerobic glycolysis, or the Warburg effect, has been observed in thousands of experiments and inspired treatments that aim to stop tumor growth by preventing cancer cells from increasing their glucose consumption. To date, this treatment approach has not proven successful in clinical trials.
Considering these clinical limitations, Christofk and Lowry set out to examine if increased glucose consumption is truly indispensable to cancer formation and growth.
They decided to approach this problem using squamous cell skin cancer as a model, as they had made two key discoveries about the nature of this cancer in recent years.
In 2011, they determined that squamous cell skin cancer, which forms in the thin, flat cells found on the surface of the skin - can originate from hair follicle stem cells. Hair follicle stem cells produce hair throughout a person's lifetime and remain mostly inactive, but spring to action during a new hair cycle, which is when new hair growth occurs. In 2017, the pair found that hair follicle stem cells are glycolytic and ramp up their glucose consumption to quickly activate and produce hair follicles.
"These findings led us to question: Are squamous cell skin cancer cells glycolytic because they are cancer cells that altered their metabolism to fuel their rapid growth, or because the cells they originated from—hair follicle stem cells—were glycolytic?" said Lowry, a professor of molecular, cell and developmental biology.
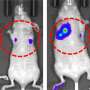
To answer this question, the team studied the progression of squamous cell skin cancer in animal models whose hair follicle stem cells had been genetically modified to limit their glucose consumption. Specifically, they de-activated a gene called lactate dehydrogenase-a, which catalyzes the final step in a cell's process of converting glucose to lactate. Deactivating this gene prevented this final step from taking place, which in turn caused the cells to dramatically reduce their glucose consumption.
The change had no effect on cancer incidence or progression. When faced with insufficient glucose for their increased needs, the cancer cells in this model simply altered their metabolism to derive energy from the amino acid glutamine.
"These findings suggest that tumors are metabolically flexible and can use nutrients other than glucose to fuel growth," said Christofk, an associate professor of biological chemistry and molecular and medical pharmacology. "Understanding all of the nutrients cancers use for growth is critical to developing drugs that can successfully target cancer's metabolism."
The team double-checked their findings by conducting a converse experiment using hair follicle stem cells that had been genetically modified to increase glucose consumption. If their initial finding was incorrect, stimulating glucose consumption would make the tumors grow faster—it did not.
"The cells still formed cancer, but they didn't do so any faster and it wasn't any more serious," said Aimee Flores, a postdoctoral fellow in Lowry's lab and a first author of the study. "The behavior and progression of the disease was quite similar to the disease observed in the model with reduced glucose consumption."
As a next step, the team will conduct experiments to determine if reducing the consumption of both glucose and glutamine can stop the growth of squamous cell skin cancers.
"If limiting cancer's intake of both of these nutrients is shown to be effective, then that points to a path toward the clinic in the form of a combination therapy," said Lowry.
There is already some evidence that a combination therapy of this kind could treat squamous cell lung cancer. David Shackelford, an associate professor in the division of pulmonary and critical care medicine and a colleague of Christofk and Lowry's at the UCLA Jonsson Comprehensive Cancer Center, found that squamous cell lung cancers metabolize glutamine when unable to increase their glucose consumption.
Shackelford and his collaborators—including Christofk—also identified two drug candidates that, when used in combination, may stop the growth of squamous cell lung cancers by reducing the uptake of both of these nutrients.
Despite these encouraging findings, the road to bringing combination therapies for squamous cell cancers to humans is a long one, cautioned Lowry. "Every drug you add to a potential treatment carries its own risks and side effects, so identifying and testing combination therapies that will be both safe and effective in humans is a long and arduous process," he said.
More information: A. Flores et al, Increased lactate dehydrogenase activity is dispensable in squamous carcinoma cells of origin, Nature Communications (2019). DOI: 10.1038/s41467-018-07857-9