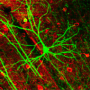
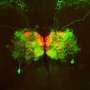
Connecting neurons in the brain
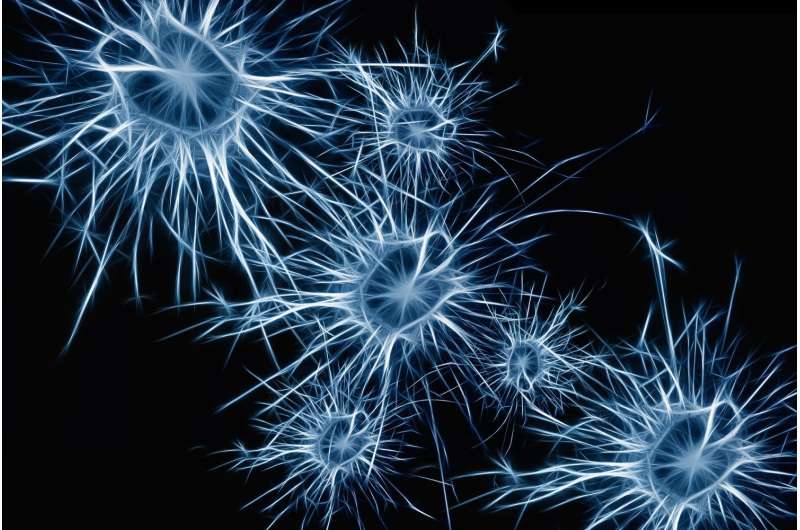
The brain consists of a large collection of interconnected neurons. How complex patterns of neuronal cells grow into functioning circuits during development has fascinated researchers for decades. A team of scientists at VIB and KU Leuven has now uncovered a new signaling mechanism in fruit flies that specifies the formation of neuronal circuits in the brain.
About 100 billion neurons form a complex and interconnected network in the brain, allowing people to generate complex thought patterns and actions. Neurons come in all sizes and shapes, but they mostly have long protrusions that connect to neighboring cells through specialized information-transmission structures called synapses.
How this intricate network takes shape during early development captivates many neuroscientists, including Prof. Dietmar Schmucker (VIB-KU Leuven) who has built a career studying neuronal wiring. "Proper brain functioning relies on very controlled branching of neuronal cell-extensions called axons and dendrites, and the correct formation of synapses at precise locations along these branches," he says. "Specifying synapse formation determines where and how many potential connections a neuronal cell is allowed to form. Therefore, controlling synapse numbers at each neuronal branch is essential for the correct formation of complex brain circuits."
A new player
Schmucker's team turned to the developing fly brain to study which molecular players control synapse formation in specific subcellular compartments. Using a genetic single-cell approach, the researchers could label and manipulate individual neuronal protrusions in the nervous system of the fruit fly, a popular model organism for neuroscientists.
"We found differences in neuronal branching and in synapse numbers at individual protrusions of neurons of the same type," explains Olivier Urwyler, a postdoc in the lab who developed this new experimental system. Urwyler, now a group leader at University Zurich, found that a phosphatase called Prl-1 was decisive for specifying where to form the highest density of synaptic connections on a given neuron.
In fruit flies, loss of Prl-1 led to defects in the formation of neuronal connections in several different circuits, suggesting that this protein phosphatase is of general importance in circuit formation. The team also identified through which signaling pathway Prl-1 exerts its function.
"Surprisingly, it turns out to be one of the most ubiquitously acting signaling pathways, the Insulin receptor/Akt/mTor pathway, required in many physiological responses, cellular growth and cancer, says Urwyler. "Restricting the subcellular protein distribution of Prl-1 to a small compartment results in this potent signaling cascade to locally boost synapse formation."
From flies to humans?
Flies that lack Prl-1 show severe locomotor problems. Interestingly, if Prl-1 is erroneously overexpressed and out of control, it can drive metastatic behavior of cancer cells.
As Prl-1 phosphatases are conserved from invertebrates to mammals, what could this imply for humans? According to Schmucker, their presence in different regions of the human brain means that Prl-1 phosphatases are poised to function in a similar way during vertebrate brain development:
"The compartmentalized restriction of Prl-1 could serve as a specificity factor to control the precise tuning of synaptic connections in human neurons as well, similar to the effects we have shown for the assembly of neuronal circuits and synapses in fruit flies."
More information: "Branch-restricted localization of phosphatase Prl-1 specifies axonal synaptogenesis domains," Science (2019). science.sciencemag.org/cgi/doi … 1126/science.aau9952