Metabolic reprogramming of branched-chain amino acid facilitates drug resistance in lung cancer
Despite the remarkable success of molecular targeted therapy in recent years, the rapid increase of drug resistance is a major obstacle to effective treatment of lung cancer.
How do lung cancer cells adapt to targeted therapy? What is the molecular basis of such adaptive behavior? Can this adaptive response be memorized by cancer cells? If so, is it heritable? The answers to these questions will provide a deeper understanding of the evolutionary process of drug resistance during molecular targeted therapy.
In a study published online in Cell Reports, research teams led by Dr. Ji Hongbin at the Institute of Biochemistry and Cell Biology of the Chinese Academy of Sciences, Dr. Zhou Caicun at the Shanghai Pulmonary Hospital, Tongji University School of Medicine, and Dr. Li Cheng at the School of Life Sciences, Peking University, revealed the important role of epigenetic regulation-mediated metabolic reprogramming in lung cancer's capacity to resist molecular targeted therapy.
The researchers found that low-dose targeted drug pretreatment allowed epidermal growth factor receptor (EGFR)-mutant lung cancer cells to adapt to subsequent high-dose drug treatment, thus exhibiting a transient drug-tolerant state. They also found that continuous stimulation with low-dose drugs reinforced this adaptive response, and eventually promoted the development of drug resistance.
Such adaptive behavior occurs not only in EGFR-mutant lung cancer, but also in ALK-rearranged lung cancer, thus indicating this phenomenon is not restricted to one lung cancer type.
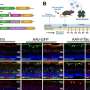
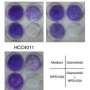
In addition, the researchers revealed the molecular basis of this adaptive response, which involves epigenetic regulation-mediated metabolic reprogramming. During the adaptation of lung cancer cells to drug therapy, the level of intracellular histone H3K9 methylation is reduced, thus up-regulating the branched-chain amino acid aminotransferase 1 (BCAT1). In this process, more glutathione is generated by BCAT1, thereby effectively eliminating detrimental oxidative stress caused by targeted therapy and eventually causing drug resistance.
Not surprisingly, correlation analyses of clinical data showed that high expression of BCAT1 in tumors is associated with an unfavorable therapeutic response to drug treatment and predicts poor patient prognosis.
However, using preclinical mouse models, the researchers demonstrated that the combination of targeted therapies with reactive oxygen species (ROS)-inducing drugs can effectively overcome lung cancer drug resistance, suggesting a potential therapeutic strategy for dealing with such drug resistance.