Protein's role in inflammation-related cancer studied
It's well established that chronic inflammation can lead to colon cancer, but the molecular mechanisms behind this association aren't fully understood. Research at Vanderbilt University Medical Center into the role that the signaling protein SMAD4 plays in this process has received funding from the National Cancer Institute.
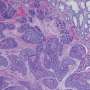
The co-principal investigators of the $2.17 million grant are Anna Means, Ph.D., research associate professor of Surgery and Cell and Developmental Biology, and R. Daniel Beauchamp, MD, John Clinton Foshee Distinguished Professor of Surgery and professor of Cell and Developmental Biology. They and colleagues linked loss of SMAD4 to inflammation-driven carcinogenesis in the colon in a study published last year in the journal Cellular and Molecular Gastroenterology and Hepatology.
They showed a complete loss of SMAD4 occurred in almost half of colitis-associated cancers.
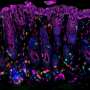
"This grant examines the role of transforming growth factor beta (TGF beta) signaling through a key transcription factor protein called SMAD4," Beauchamp said. "What we found was that induced loss of the SMAD4 protein in colonic epithelial cells in mice resulted in increased inflammatory gene expression in those epithelial cells and an associated increase in inflammatory cell infiltration into the colonic stroma around the colonic glands.
"Conversely, we found that with intact SMAD4, TGF beta could inhibit or extinguish inflammatory responses in colon epithelial cells. Our data suggest strongly that epithelial cell anti-inflammatory action of TGF beta signaling through SMAD4 is central to the tumor suppressor function of this pathway. Consistent with these observations in mice and colon epithelial cell cultures, we found that there is frequent loss of SMAD4 protein in human ulcerative colitis-associated carcinoma.
"This grant helps us pursue the mechanisms underlying the links between TGF beta and SMAD4 signaling, inflammation and tumor suppression," Beauchamp said.
The grant, which is for a five-year period, supports research that could possibly lead to prevention measures for some colorectal cancers, especially for patients at higher risk.
"People with inflammatory bowel disease clearly have a predisposition to develop cancer," Means said.
"We think we have found a key mechanism that may regulate this progression from inflammatory disease to cancer. What we're trying to do with this grant is understand mechanistically what happens. How does SMAD4 regulate this transition? If we can understand exactly what it is doing, can we target that so we can maybe prevent cancer from occurring in these high-risk patients?"
Key collaborators on the research include Ken Lau, Ph.D., associate professor of Cell and Developmental Biology, Qi Liu, Ph.D., assistant professor of Biomedical Informatics, and Chanjuan Shi, Ph.D., professor of Pathology, Microbiology and Immunology.
While their work will focus on colorectal cancer, the research has possible relevance for other cancers of the alimentary tract, including pancreatic cancers and head and neck cancers that are inflammation-related, Beauchamp said.