Alzheimer's amyloid build-up affects certain parts of brain cells more than others in mice

A protein that is linked with Alzheimer's Disease has been found to be more likely to affect certain parts of brain cells that send messages than other parts of the cells.
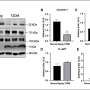
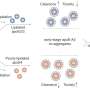
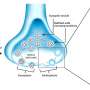
In a new study published in Scientific Reports, the team observed how amyloid beta (Aβ), a protein which accumulates in the brain of people with Alzheimer's disease, affected the parts of nerve cells, the synapses, in mice which are responsible for the communication with each other.
The team found that Aβ build-up had more effect on the axons, the part of the synapse involved in sending signals, than the dendrites, the area responsible for receiving them. The finding will help the development of future work on Alzheimer's Disease to target particular parts of the brain cells.
Dr. Johanna Jackson, the lead researcher of the paper, from Imperial College said:
"This study demonstrates how different parts of the synapse, the part of the cell which communicates with other cells, are affected differently by amyloid pathology in this model. When studying and developing therapies for diseases which include synapse loss, such as Alzheimer's Disease, both parts of the synapse must be taken into account, which is often not the case."
Dr. Francesco Tamagnini, Lecturer in Pharmacology at the University of Reading said:
"This study suggests that the development of Alzheimer's Disease may begin to affect a very specific part of our brain cells as the amyloid pathology that contribute to the disease is more likely to build up in different areas.
"As we don't tend to see the early damage that Alzheimer's causes on our brains, this new discovery about the part of our synapses that are affected by the pathology that builds up is important. It means that we can now look more closely at that part of the brain cell earlier, and develop drugs that can help combat the disease."
More information: Terri-Leigh Stephen et al. Imbalance in the response of pre- and post-synaptic components to amyloidopathy, Scientific Reports (2019). DOI: 10.1038/s41598-019-50781-1