December 27, 2019 feature
How signalling proteins affect wound healing
What do a scraped knee, a paper cut, or any form of surgery have in common? The short answer is a wound in need of healing—but the long answer lies in a series of biological activities that allow tissues to repair themselves.
Questions about wound healing have been asked for more than 1,000 years. Now, scientists have begun decoding how signaling proteins are intimately involved in a process that transforms an open wound into remodeled healthy tissue.
While knowing more about simple wounds is important, the research underway at Virginia Commonwealth University also has a larger aim—helping to solve extraordinarily complex medical problems. Inflammatory disorders such as sepsis, anaphylaxis and trauma-induced coagulopathy might eventually be aided by research into wound repair. Trauma-induced coagulopathy refers to the blood's inability to coagulate—form clots. The impairment, caused by severe trauma, can lead to prolonged or excessive bleeding, which can prove fatal.
Wounds heal through a dynamic process that involves various molecular pathways and cell types that are coordinated to facilitate healing in a timely and orderly manner, Dr. Charles E. Chalfant and colleagues write in the journal Science Signaling.
"One of the key players in this orchestra is a group of molecules called eicosanoids, which are derived from fatty acids, and control both inflammation and the migration of cells around the wound site," reported Chalfant, who heads the lipid signaling research laboratory at Virginia Commonwealth University.
Eicosanoids not only are fatty-acid-derived molecules, as Chalfant and his colleagues have found, they are also signaling proteins. Signaling molecules are critical components of cells that allow short and long distance communications. Imagine a microscopic network that is roughly comparable to a telephone system through which calls are relayed and received.
As it turns out, eicosanoids steer two key stages of wound repair, Chalfant his team of investigators discovered in a groundbreaking investigation that has implications for surgical patients, accident victims, and possibly one day for those with potentially deadly inflammatory disorders. H. Patrick MacKnight, a colleague of Chalfant at Virginia Commonwealth University and the molecular biology division of the University of South Florida in Tampa, served as first author on the new report.
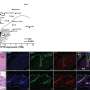
Wound repair, the team revealed, advances through distinct phases, which includes inflammation followed by proliferation and tissue remodeling. The team reported in Science Signaling that fibroblasts infiltrate the wound, proliferate, and secrete collagen, which is remodeled as the epithelium regenerates.
In their observations of laboratory animal models—mice that were assessed on a variety of wound-repair parameters—the scientists were able to document each step. They also noted the importance of eicosanoid signaling, which is crucial to the repair process.
Eicosanoids have to be produced by the body, according to Chalfant and his colleagues. They say the sphingolipid, ceramide 1-phosphate, binds to and activates group IVA cytosolic phospholipase to stimulate the production of eicosanoids.
"Because eicosanoids are important in wound healing, we examined the repair of skin wounds in knockout mice," Chalfant wrote. The knockout (KO) mice were compared with their "knock-in" (KI) counterparts. The knockouts lacked the protein cPLA2α, but the knock-ins were endowed with it.
The wound closure rate was not affected in the KO or KI mice, but wound maturation was enhanced in the KI mice. Their wounds displayed increased infiltration of dermal fibroblasts, increased wound tensile strength and several other noteworthy differences compared with their KO counterparts.
The research is part of active area of inquiry in Chalfant's laboratory, which is examining how lessons learned about inflammatory processes in wound repair can help shed new light on more complex medical conditions.
Eicosanoids, for example, are known mediators of inflammatory responses. The molecules underlie the pathogenesis of sepsis and other major medical disorders.
With sepsis mortality exceeding more than 200,000 people in the United States annually, it is time to look at it from a fresh perspective, according to Chalfant's laboratory mission statement. Knowledge gained from wound repair research is a step in a new direction, he says.
More information: H. Patrick MacKnight, et al. The interaction of ceramide 1-phosphate with group IVA cytosolic phospholipase A2 coordinates acute wound healing and repair, Science Signaling (2019) DOI: 10.1126/scisignal.aav5918
© 2019 Science X Network