Scientists find new way to block cancer-causing HPV virus
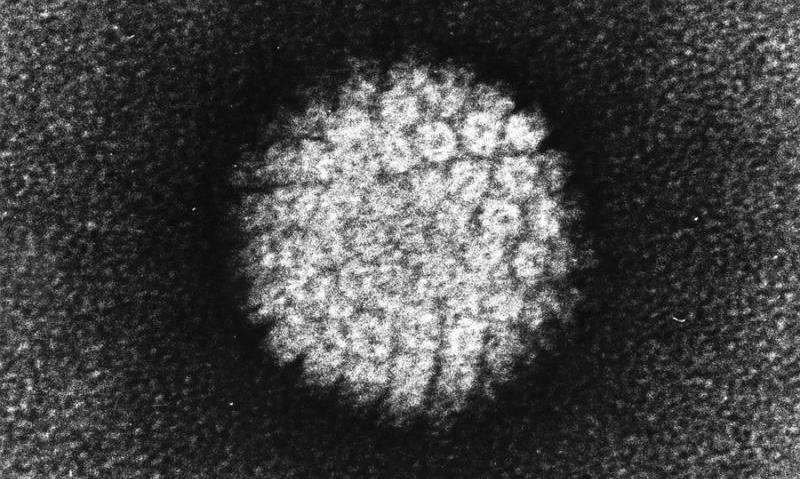
The human papillomavirus (HPV) is the main cause of several cancers, including cervical cancer, which kills almost 300,000 women around the world each year. Although vaccines offer a proven first line of defense against HPV infection, researchers continue to look for additional options to guard against the virus.
In a new study published today in the Proceedings of the National Academy of Sciences, Yale Cancer Center (YCC) researchers have demonstrated in principle a new biological approach that can stop HPV infection. This method may eventually aid in treating not only HPV, but other viruses, as well as non-viral diseases that are currently thought to be "undruggable," said the researchers.
"We show that very short peptides [fragments of a protein] can block the HPV virus from infecting cells," said senior author Daniel DiMaio, M.D., Ph.D., deputy director of YCC, the Waldemar Von Zedtwitz Professor of Genetics, and professor of molecular biophysics and biochemistry and of therapeutic radiology. "This research confirms our model for how HPV infects cells. It also shows that the intracellular trafficking of a virus could be the target for a new anti-viral approach."
HPV is carried into the cell by a membrane-bound sac called an endosome. An HPV protein known as L2 contains a segment known as a "cell-penetrating peptide" that sticks through the membrane of the endosome into the cell's interior. There, a sequence of L2 next to the cell-penetrating peptide binds to a cell protein called retromer. Retromer then delivers the virus into a cellular transport mechanism known as the retrograde pathway that drops off the virus in the nucleus, where it can begin making copies of itself.
Previous research by DiMaio's lab found that the core machinery of the cell-penetrating peptide is surprisingly short. Peptides are composed of amino acids, and a sequence of only six amino acids was needed for the peptide to penetrate cell membranes, while a sequence of only three amino acids was required to bind to the retromer protein.
"We realized that we could synthesize a short peptide that should be enough to get through the cell membrane, bind the retromer and block infection, so we decided to test that," DiMaio said. "The first peptide we tried worked."
When the investigators added the synthesized cell-penetrating peptides into a culture medium of human cells, they saw that the peptides did enter the cytoplasm and bind to the retromer. When the scientists then infected the cells with HPV, the virus could no longer bind to the retromer and leave the endosome because the retromer was tied up by the peptide, and infection was blocked.
The Yale researchers demonstrated that this peptide inhibition persists even after the peptides are removed. "We don't actually know how long the peptide is active, but the effect may be irreversible," DiMaio added. "It also looks like the virus disappears from the cell. The cell has some way to sense that the infection is not proceeding normally, so it gets rid of the virus."
In follow-up experiments conducted with colleagues at the University of Wisconsin, the scientists showed that this cell-penetrating peptide also inhibited HPV infection in mice. This basic research may point toward new types of anti-HPV treatments, which are needed, DiMaio said.
Although vaccines will always be the best foundation to prevent HPV infections, DiMaio said, "the vast majority of people worldwide are not vaccinated, especially in the developing world where most cases of cervical cancer occur." Additionally, current vaccines don't guard against all strains of HPV, he said.
More information: Pengwei Zhang et al. Cell-penetrating peptide inhibits retromer-mediated human papillomavirus trafficking during virus entry, Proceedings of the National Academy of Sciences (2020). DOI: 10.1073/pnas.1917748117