Mechanisms of mechanical-stretch-induced skin expansion revealed
The capacity of the skin to expand by mechanical stretching has been used for decades in plastic and reconstructive surgery to generate an excess of skin that can be used to repair birth defects, damaged tissues, and breast reconstruction after mastectomy. The cellular and molecular mechanisms by which skin respond to mechanical stretching remain unknown.
Researchers lead by Pr. Cédric Blanpain—WELBIO investigator, director of the Laboratory of Stem Cells and Cancer, Faculty of Medicine, Université libre de Bruxelles, in collaboration with Pr. Benjamin D. Simons, University of Cambridge, demonstrated that stretching induces skin expansion via self-renewal of epidermal stem cells and uncovered the signaling pathways responsible for stretching induced stem cell activation and renewal at the single cell resolution.
The skin epidermis is an essential barrier protecting animals against infections and water loss. As the skin is exposed to mechanical stress, it must resist and by adapting its size while maintaining its barrier functions. These properties are used by plastic surgeons to generate extra skin for reconstructive surgery. To this end, surgeons use an inflatable "skin expander" that they insert below the skin and inflate, causing the expansion of the overlaying skin. Although this technique has been used for decades in medicine, very little is known about the mechanisms by which the mechanical force of the skin expander lead to activation of skin stem cells and the production of an excess of skin.
In this new study published in Nature, Mariaceleste Aragona and colleagues define the clonal dynamics and the molecular mechanisms that lead to stem cell activation and skin expansion at single-cell resolution.
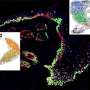
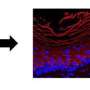
They used state-of-the-art genetic mouse models to study the reaction of the epidermal stem cells to mechanical stretching. Specifically, they performed genetic lineage tracing that allowed them to mark the stem cells, which reside at the top of the cellular hierarchy in the skin, and follow the fate of the stem cell progeny over time. They coupled these analyses with single-cell sequencing in collaboration with Pr Thierry Voet—KULeuven—that analyze the molecular identity of each individual cell providing a deep resolution of different cellular states that accompanied skin expansion.
Interestingly, they found that only specific stem cells were changing their behavior and cell fate in response to mechanical stretching. "It was particularly exciting to observe that not all cells respond equally to mechanical perturbations and to identify for the first time the molecular signature that characterize these specific stretched skin stem cells," says Mariaceleste Aragona, the first author of the study.
In collaboration with Pr. Benjamin D Simons, University of Cambridge, they developed a mathematical model of how the skin expands upon stretching. During stretch-mediated skin expansion, a transient increase in stem cell self-renewing division allows the tissue to maintain its cellular composition and organization, while expanding the pool of cells that will mediate the tissue expansion. These data demonstrate the plastic and resilient nature of stem cells, which is fundamental to allow the tissue to respond to its environment and expand when needed while maintaining the vital barrier functions of the skin.
This study dissects the spatiotemporal cellular and molecular mechanisms that control the behavior of skin stem cells in response to mechanical stretch and delineates step-by-step the molecular components that allow the stem cells to sense and mediate the response to stretch and induce tissue expansion.
"These new findings, not only uncover how the skin reacts to mechanical perturbations through the activation of specific population of stem cells, but also unravel the molecular players responsible for stem cell mechanotransduction in a clinically relevant context. We hope that the understanding of these new mechanisms will be exploited in the future to stimulate tissue expansion and wound healing," says Cédric Blanpain, the senior author of the Nature paper.
More information: Mariaceleste Aragona et al. Mechanisms of stretch-mediated skin expansion at single-cell resolution, Nature (2020). DOI: 10.1038/s41586-020-2555-7