Pacify the protein and win over a disease
The interests of Karolina Mikulska-Ruminska, Dr., from the Department of Biophysics in the Institute of Physics, NCU focus on widely understood biological structures, especially proteins. In her work, she applies calculation methods in computer simulations. During her postdoctoral internship in the group of Ivet Bahar, Prof. at the University of Pittsburgh, she already began her cooperation with medical doctors from Los Angeles and the researchers from the University of Pittsburgh.
The latest effects of her scientific inquiries can be read about in the article "Recruitment of pro-IL-1α to mitochondrial cardiolipin, via shared LC3 binding domain, inhibits mitophagy and drives maximal NLRP3 activation," which has just been published in PNAS. Doctor Mikulska-Ruminska and Jargalsaikhan Dagvadorj from Cedars-Sinai Medical Center in Los Angeles are the first authors of the publication. The article refers to the mechanisms of the activities of the immune system under inflammatory conditions.
"One of the main objectives of our research was to define what role is played by the protein prointerleukin-1 alpha (pro-IL-1α). It is known that the mature form of this pro-inflammatory cytokine (IL-1α) is responsible for the formation of inflammatory states, fever and sepsis. It was a huge discovery to find out that the same protein in an inactive form of precursor (pro-IL-1α) plays such a key role in regulating the response of the immune system," explains Dr. Mikulska-Ruminska.
Dangerous inflammation
Interleukins (IL) are a group of cytokines—that is, proteins—which take part in inflammatory processes of the immune system. The proteins are numbered—a few dozen have been classified. Dr. Mikulska-Ruminska got her interest in the first group: IL-1. This is a collective term which refers to the cytokines of key importance in inflammation processes. They are secreted in response to various types of antigens, for instance of viral, bacterial, or fungal origin. Out of 10 different strains, one of the most significant is the already mentioned IL-1α, which most often occurs as a membrane cell and it interacts with the neighboring cells and IL-1β. It is these cytokines that have become the focus of the examination of the Polish researcher and American scientists.
"At this point, it is necessary to mention the inflammasome NLRP3. This is a structure within a cell which consists of many specialized proteins. In a healthy cell it remains inactive. It becomes activated when the organism is endangered with, for instance, the presence of microorganisms or cells which appeared due to the damage of tissues, and metabolic disorders," explains Dr. Mikulska-Ruminska. Active inflammasomes NLRP3 transform the interleukin 1 beta (IL-1β) and interleukine18 (IL-18) into their active forms, due to which an inflammatory state is generated in the organism.
Obviously, there is quite a numerous group of inflammations which are necessary for the organisms, for instance to eliminate bacteria or viruses. A chronic state, however, leads to many dangerous diseases. The best example of this is so-called "cytokine storm," which is a hyperreaction of the immune system, a serious problem in the most severe cases of COVID-19, explains Dr. Mikulska-Ruminska. Hyperactivity of NLRP3 triggers off numerous pathological conditions, including atherosclerosis, arthritis, inflammatory bowel disease or type 2 diabetes.
A lot of scientists also find a connection between a chronic inflammatory state and Alzheimer's disease, which is a progressive neurodegenerating disease that leads to gradual atrophy of cognitive and memory functions. Although the pathophysiological mechanism of the disease has not yet been entirely discovered, researchers believe it is related to the activation of NLRP3 inflammasome.
"Our research reveals the mechanism due to which cells modulate the activation of NLRP3 inflammasome. This is very crucial as it is connected with the possible therapeutic application, for example while treating the above-mentioned diseases," says Dr. Mikulska-Ruminska. "We have found that the cells which lack pro-IL-1α are characterized by a decreased activity of NLRP3 inflammasome and caspase-1 (an element of the inflammasome which activates cytokines). This leads to a lesser release of IL-1β and decreased damage of mitochondria. The role of pro-IL-1α is therefore extremely important—it regulates the activation of the inflammasome, and thus is a key protein to initiate an inflammatory state."
Competing proteins
The scientists have also become interested in one more issue. Dr. Mikulska-Rumiska has discovered that the fragment IL-1α (so called signal peptide) is extremely similar to another protein—LC3b, which plays a key role in purifying the organism from damaged or redundant cells. The sequences of both of them are almost identical.
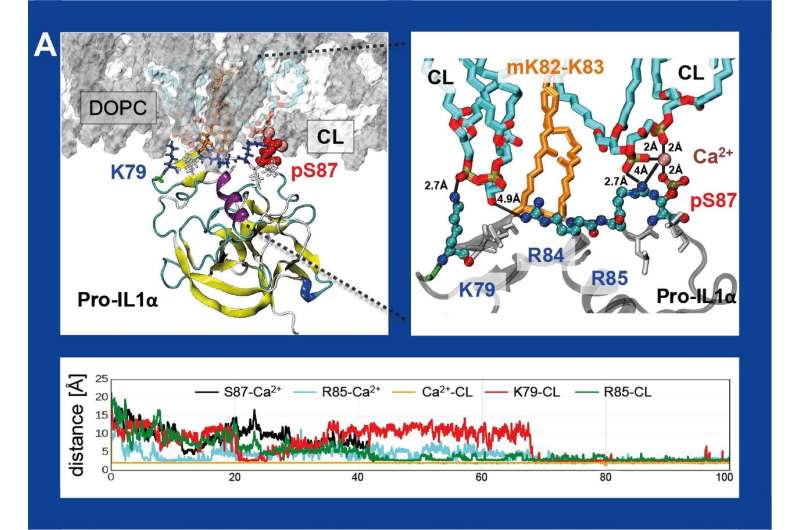
"We have shown that pro-IL-1α interacts in the mitochondrial membrane with cardiolipin. Cardiolipin is an important phospholipid which normally binds with another protein—LC3b and, at the same time, sends a signal 'eat me' to eliminate call," explains Dr. Mikulska-Ruminska.
Simulations of the molecular dynamics for such a set have shown what structural elements are the key elements for interactions with cardiolipin. What is more, it has turned out that the same structural elements are present in the protein LC3b.
"These proteins may compete with each other, which may trigger off serious consequence," explains Dr. Mikulska-Ruminska. "When pro-IL-1α in, for instance, a damaged cell connects with cardiolipin, LC3b is not able to perform its job, which is to communicate to the organism that it should be 'thrown out.' This happens because cardiolipin gets intercepted by pro-IL-1α and can no longer interact with LC3b."
This discovery may also contribute to developing new strategies of treatment of inflammatory states responsible for the development of numerous diseases.
More information: Jargalsaikhan Dagvadorj el al., "Recruitment of pro-IL-1a to mitochondrial cardiolipin, via shared LC3 binding domain, inhibits mitophagy and drives maximal NLRP3 activation," PNAS (2020). www.pnas.org/cgi/doi/10.1073/pnas.2015632118