Experimental treatment appears to subdue type 1 diabetes in laboratory mice

An experimental treatment can essentially reverse type 1 diabetes in certain types of laboratory mice, according to a series of studies led by University of Utah Health scientists. An injection of the therapeutic agent converts cells that normally control glucose production into ones that generate insulin.
The researchers say giving the animals a single dose of a human antibody that suppress the actions of glucagon, a hormone involved in glucose regulation, sparked a remarkable transformation in the pancreas, leading to a nearly 7-fold increase in insulin cell mass and the suppression of diabetic symptoms.
"These animals go from requiring insulin injections to never requiring a diabetes treatment again. They maintain normal blood glucose long after we stop the treatment," says William L. Holland, Ph.D., the study's corresponding author and a U of U Health assistant professor of Nutrition and Integrative Physiology. "What this implies for millions of people who have type 1 diabetes is that there is a chance we could eventually regenerate their own insulin-producing capacity, restore their normal blood glucose balance, and hopefully help them make progress toward being free of the disease."
However, the researchers caution they are still far from this goal. "This research is certainly promising, but it is likely just the first step of many before we can determine if this approach will work in humans with type-1 diabetes," Holland says. "We have a long, long way to go."
The study appears in Proceedings of the National Academy of Sciences (PNAS). In addition to U of U Health, researchers from Vanderbilt University Medical Center, Baylor College of Medicine, Lilly Research Laboratories, the University of Texas Southwestern Medical Center, the Veteran's Administration in Dallas and the Juvenile Diabetes Research Foundation (JDRF) contributed to this effort.
Glucagon and insulin are produced by groups of cells in the pancreas known as islets of Langerhans. These hormones work in tandem to keep blood glucose under control. However, Type 1 diabetes disrupts that process by triggering the body's immune system to attack and destroys insulin-producing cells. As a result, most people with Type 1 diabetes rely on insulin injections or pumps to survive.
"By the time, individuals are diagnosed with Type 1 diabetes, they've probably lost 90% of the cells that produce insulin in their bodies," says E. Danielle Dean, Ph.D., a study co-author and assistant professor of medicine at Vanderbilt University Medical Center. "So, in order for a person to achieve glucose control without treating themselves with insulin, you've got to convince the cells in the body that produce insulin to increase their numbers."
To determine if that can happen, Holland, Dean and colleagues induced diabetes in mice, triggering the death of insulin-producing beta cells in the pancreas. When these same mice were given a human monoclonal antibody called Ab-4 that blocks glucagon binding in the liver, their blood glucose levels normalized and circulating blood levels of insulin were restored.
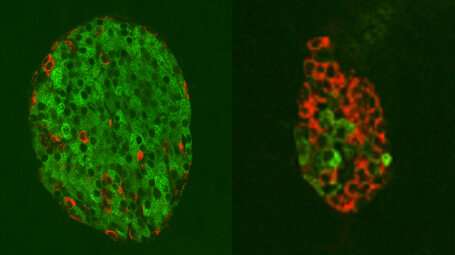
Digging deeper, the researchers tracked glucagon-producing alpha cells using a fluorescent protein marker that glows red. The increased number of glowing red cells were also producing insulin suggesting that these glucagon producing cells had started making insulin instead. These initial studies were performed in mice that were able to regenerate insulin-producing cells without interference from the immune system.
Next, the scientists turned their attention to non-obese diabetic (NOD) mice. These animals spontaneously develop diabetes because their overly aggressive immune systems readily provoke beta (insulin) cell death. This condition closely resembles Type 1 diabetes in humans. However, treatment with Ab-4 restored insulin production and regeneration of abundant numbers of insulin producing cells in the pancreas.
"It is a very intriguing finding that our treatment protects and restores insulin-producing beta cells even in the presence of a persistant immune response" Holland says. "It appears that the immune cells can no longer kill the new beta cells."
However, many potential therapies that increase beta cell mass in mice fail to have the same effect in human islets or in patients. To see if they could overcome that, the researchers grafted human islets into immunodeficient, diabetic mice. They then selectively killed mouse beta (insulin) cells, leaving only the human islets in place. Treating these mice with Ab-4 enhanced glucose control and increased the amount of circulating human insulin in their bloodstreams, confirming a benefit to human islets
These effects were still detectable 40 days following treatment.
Moving forward, the researchers are beginning to investigate how alpha (glucagon) cells convert into insulin-producing cells and avoid being damaged by the immune system.
More information: May-Yun Wang el al., "Glucagon blockade restores functional β-cell mass in type 1 diabetic mice and enhances function of human islets," PNAS (2021). www.pnas.org/cgi/doi/10.1073/pnas.2022142118