Myositis-specific autoreactive T cells are pathogenic for dermatomyositis
Dermatomyositis is an idiopathic inflammatory myopathy that has been regarded as an autoimmunity-based disorder, although its pathogenesis remains unclear. In this study, researchers from the University of Tsukuba used a mouse model to identify a mechanism by which dermatomyositis may develop in humans. The animal model and findings can be used to better understand the disease and develop disease-specific treatments.
Dermatomyositis belongs to a group of idiopathic inflammatory myopathies that are associated with the presence of specific autoantibodies in patient sera. Multiple myositis-specific autoantibodies, which target proteins ubiquitously expressed in the nucleus or cytoplasm, have been described. One of these autoantibodies is specific for transcriptional intermediary factor 1γ (TIF1γ).
"Autoimmune diseases constitute a difficult challenge for patients and doctors," says lead author of the study, Professor Naoko Okiyama. "Not only are the diagnosis and treatment of autoimmune diseases clinically difficult, but it is often unclear why and how the diseases developed. In this study, we investigated the role of a specific autoantibody present in the sera of patients with dermatomyositis, the anti-TIF1γ antibody, in the pathogenesis of the disease."
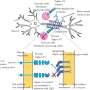
To achieve their goal, the researchers injected TIF1γ protein into normal mice, as well as several mouse models lacking proteins that play distinct roles in the body's immune response. The researchers' aim was to determine which part of the immune system contributes to the development of dermatomyositis. In normal mice, injection of TIF1γ resulted in the production of TIF1γ-specific T cells and anti-TIF1γ autoantibodies, thereby inducing myositis. In this myositis, CD8+ T cell infiltration into muscle cells resulted in muscle fiber wasting. Generally, CD8+ T cells are important for eliminating infected cells, cancer cells, and other damaged cells.
Importantly, the disease severity was significantly reduced when TIF1γ was injected into mice that lacked the ability to present antigens to CD8+ T cells. Adoptive transfer of CD8+ T cells, but not antibodies, collected from TIF1γ-treated mice led to myositis in recipient mice. This suggests that autoreactive CD8+ T cells against TIF1γ contribute to the pathogenesis of dermatomyositis, while autoantibodies against TIF1γ are simply non-pathogenic clinical diagnostic markers.
"In contrast to existing animal models of experimental myositis, which use muscle-specific antigens, our results show that autoreactive T cell-mediated autoimmunity to TIF1γ may play a causal role in dermatomyositis. This new experimental model may be a new tool to further investigate the disease and develop new therapeutics against dermatomyositis," says Professor Okiyama.
More information: Naoko Okiyama et al. Immune response to dermatomyositis-specific autoantigen, transcriptional intermediary factor 1γ can result in experimental myositis, Annals of the Rheumatic Diseases (2021). DOI: 10.1136/annrheumdis-2020-218661