Mechanism for development of rare colorectal cancer subtype identified
Researchers from Rutgers Cancer Institute of New Jersey, the state's only National Cancer Institute-designated Comprehensive Cancer Center, recently discovered a mechanism to explain what drives the formation of mucinous colorectal adenocarcinoma (MAC), a rare subtype of colorectal cancer (CRC), and has also identified the genes responsible for the regulation of this mechanism. The OnlineFirst version of this work was published August 20 in Cancer Research .
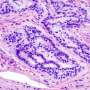
According to the American Cancer Society, colorectal cancer (CRC) is the third leading cause of cancer-related death in men and women in the United States. MAC is a distinct and aggressive form of CRC, accounting for 10 to 15 percent of patients (Nat Rev Clin Oncol, June 2016). It is characterized by the excess secretion of the protein known as mucin and has an entirely different molecular signature—or biological behavior—than classic CRC. What drives the formation of MAC tumors is currently unknown and underexplored.
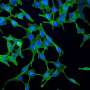
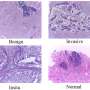
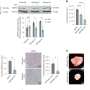
Using mouse models, investigators in this study demonstrated that loss of a tumor suppressor protein known as FILIP1L induces colonic epithelial hyperplasia and mucin secretion. They also demonstrated that FILIP1L stimulates degradation of a molecular chaperone protein known as PFDN1. This breakdown results in abnormal stabilization of PFDN1 leading to elevated mucin secretion and defects in cell division in colon cancer cells. These defects mirror the same characteristics that are seen in the aggressive form of MAC. The authors note these findings strongly implicate FILIP1L as an essential regulator of MAC tumor development.
"Decreased production of FILIP1L is associated with chemotherapy resistance and poor prognosis in ovarian and colon cancers. Elucidating the driver behind the development of mucinous colorectal adenocarcinoma gives researchers the ability to explore ways to target this mechanism and develop much needed therapeutics for this particular patient population," says the study's lead author Mijung Kwon, Ph.D., Rutgers Cancer Institute researcher and assistant professor of medicine at Rutgers Robert Wood Johnson Medical School.
"Our lab already has published evidence (PMID: 18794120, 21693594, 24327474, 24340050, 25200207 and 27776341) that FILIP1L plays a role in other cancer types such as pancreas, lung, breast, ovarian and prostate. Identifying an association between FILIP1L and mucinous colorectal adenocarcinoma further enhances our understanding of the role this novel tumor suppressor plays in the development of these cancers," notes Rutgers Cancer Institute of New Jersey Director Steven K. Libutti, MD, FACS, who is also senior vice president of oncology services at RWJBarnabas Health, vice chancellor for cancer programs for Rutgers Biomedical and Health Sciences, and senior author of the work.
The authors are continuing to explore this mechanism in both human tumor samples and in evolving mouse models in the hope that their work can lead to novel treatment and diagnostic approaches.
More information: Mijung Kwon et al, FILIP1L loss is a driver of aggressive mucinous colorectal adenocarcinoma and mediates cytokinesis defects through PFDN1, Cancer Research (2021). DOI: 10.1158/0008-5472.CAN-21-0897