Scientists reveal how vitamin A enters immune cells in the gut
Immunologists and geneticists at UT Southwestern Medical Center have discovered how vitamin A enters immune cells in the intestines—findings that could offer insight to treat digestive diseases and perhaps help improve the efficacy of some vaccines.
"Now that we know more about this important aspect of immune function, we may eventually be able to manipulate how vitamin A is delivered to the immune system for disease treatment or prevention," said Howard Hughes Medical Institute Investigator Lora Hooper, Ph.D., Chair of Immunology at UT Southwestern.
Vitamin A—a fat-soluble nutrient that the body converts to a molecule called retinol and then to retinoic acid before it can be used—is important for every tissue in the body, said Dr. Hooper, Professor of Immunology, Microbiology, and in the Center for the Genetics of Host Defense at UT Southwestern. It's particularly crucial for the adaptive immune system, a subset of the broader immune system that reacts to specific pathogens based on immunological memory, the type formed by exposure to disease or vaccines.
Although researchers knew that some intestinal immune cells called myeloid cells can convert retinol to retinoic acid, how they acquire retinol to perform this task was a mystery, said Dr. Hooper, whose lab investigates how resident intestinal bacteria influence the biology of humans and other mammalian hosts.
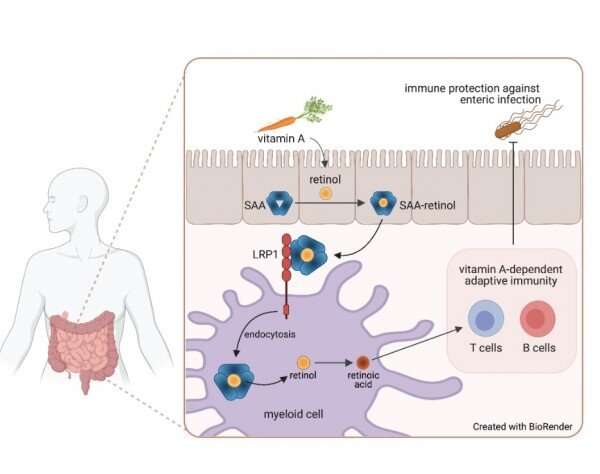
Lead author Ye-Ji Bang, Ph.D., a postdoctoral fellow in the Hooper Lab, and colleagues focused on serum amyloid A proteins, a family of retinol-binding proteins that some organs produce during infections. They used biochemical techniques to determine which cell surface proteins they attached to, and identified LDL receptor-related protein 1 (LRP1).
LRP1 was discovered more than 30 years ago by fellow UT Southwestern researcher Joachim Herz, M.D., Director of the Center for Translational Neurodegeneration Research and Professor of Molecular Genetics, Neurology, and Neuroscience. The Herz lab focuses on the molecular mechanisms by which the members of the LDL receptor gene family function as signaling and endocytic receptors in the brain and vascular wall. The discovery of the LDL receptor, made at UT Southwestern, helped earn Michael Brown, M.D., and Joseph Goldstein, M.D., the 1985 Nobel Prize in Physiology or Medicine.
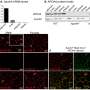
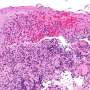
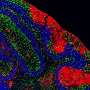
Drs. Bang, Hooper, Herz, and colleagues showed that LRP1 was present on intestinal myeloid cells, where it appeared to shuttle retinol inside. When the researchers used genetic techniques to delete the gene for this receptor in mice, preventing their myeloid cells from taking up the vitamin A derivative, the adaptive immune system in their gut virtually disappeared, said Dr. Hooper. T and B cells and the molecule immunoglobulin A, critical components of adaptive immunity, were significantly reduced. Researchers then compared the response to Salmonella infection between mice with LRP1 and those without. Those missing the receptor were quickly overcome by the infection.
The findings, published in Science, suggest that LRP1 is the vehicle by which retinol enters myeloid cells. If researchers can develop a way to inhibit this process, said Dr. Hooper, it could tamp down the immune response in inflammatory diseases that affect the intestines, such as inflammatory bowel disease and Crohn's disease. Alternatively, finding a way to enhance LRP1 activity could boost immune activity, making oral vaccines more effective.
Other UTSW researchers who contributed to this study include Zehan Hu, Yun Li, Sureka Gattu, Kelly A. Ruhn, and Prithvi Raj.
More information: Ye-Ji Bang et al, Serum amyloid A delivers retinol to intestinal myeloid cells to promote adaptive immunity, Science (2021). DOI: 10.1126/science.abf9232