New treatment for inflammatory bowel disease
Opioids are a class of substances that control sensations such as pain and emotions in animals. While plant-derived opioid narcotics such as morphine are the most well-recognized, other opioid molecules like endorphins can also be synthesized by the body or artificially developed in laboratories. Opioids exert their actions by binding to opioid receptors present on the surface of cells. While opioid receptors were earlier thought to be limited to the central nervous system (CNS), their recent discovery in other parts of the body has prompted questions about their effects in other parts of the body, including the immune system.
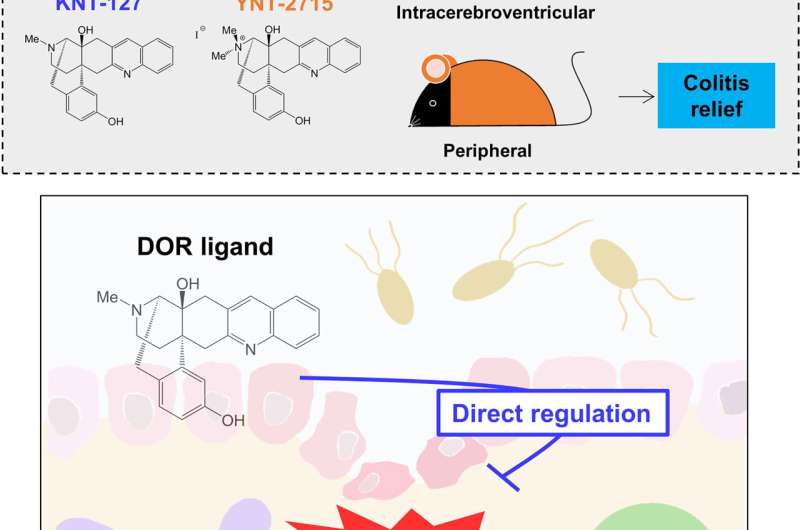
In a recent study published in Frontiers in Immunology, Prof. Chiharu Nishiyama, Kazuki Nagata, and Ayumi Okuzumi from the Tokyo University of Science and Prof. Hiroshi Nagase from the University of Tsukuba attempted to understand the effects of opioids on the immune system. They tested the effects of KNT-127—an artificially synthesized opioid that activates delta opioid receptors—on immune responses in live animal and cell culture experiments.
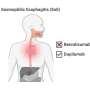
When mice with inflammatory bowel disease (IBD) were treated with KNT-127, they showed a reduction in the severity of colitis—a form of colon inflammation—indicated by lower weight loss and colon atrophy and improved disease activity scores. Similar results were also obtained in a recovery model, confirming the beneficial effects of KNT-127 against colonic inflammation.
Although these results were promising, an important caveat still loomed. "Before proceeding with additional experiments, we had to rule out the role of CNS opioid receptors in the anti-inflammatory effects of KNT-127," says Prof. Nishiyama, the lead researcher on the study. To address this, the researchers performed similar experiments with YNT-2715, a peripheral KNT-127 that cannot cross over from the blood to the brain. The results were similar to those observed with KNT-127, confirming that its anti-inflammatory effects were indeed CNS-independent.
Encouraged by this, the group examined other immune-related effects of KNT-127 treatment in the colitis model. They found that during disease progression, the opioid reduced the serum levels of IL-6, a pro-inflammatory factor, while also decreasing the number of macrophages in the mesenteric lymph nodes (MLNs). Interestingly, they also observed an increase in the number of regulatory T cells (Tregs) in MLNs. Together, their results showed that KNT-127 suppresses the inflammation caused by macrophages during disease progression and enhances the anti-inflammatory response due to Tregs during recovery.
Finally, to understand the direct effects of KNT-127 on immune cells, the researchers performed in vitro experiments in which they treated macrophages derived from bone marrow or T cells from spleen with the drug. The results were consistent with those from animal experiments, revealing increased secretion of the pro-inflammatory signals as well as enhanced development of Tregs in response to KNT-127 treatment.
Altogether, the findings demonstrated that KNT-127 can directly act on immune cells and reduce the severity of inflammation, making it a good candidate for the treatment of IBD. "Several people around the world suffer from diseases related to colon inflammation, and so far, optimal treatment strategies are lacking. Our findings show that KNT-127 and other activators of opioid receptors could be promising therapeutic options for such diseases," says Prof. Nagase, the chief drug developer behind the synthetic opioid, while also cautioning of the road ahead. "Of course, before these drugs are used clinically, additional experiments will be required to elucidate how they exert their immunomodulatory functions and what their effects on other immune diseases are."
Nevertheless, Prof. Nishiyama and her team are confident that their study represents an important milestone, not only towards the treatment of IBD but also towards our understanding of the "brain-gut axis"—the interrelationship between brain and gut function—which has received increasing attention in recent years.
"Today, we know that poor mental health has physical manifestations. For example, stress worsens inflammation in the gut, which in turn affects the health of the brain. Our results on the immune-related effects of opioids, which commonly act on the brain, is a step toward unraveling the biological mechanisms that govern the reciprocative relationship of gut health and the immune system with the CNS," says Prof. Nishiyama.
More information: Kazuki Nagata et al, Delta Opioid Receptor Agonists Ameliorate Colonic Inflammation by Modulating Immune Responses, Frontiers in Immunology (2021). DOI: 10.3389/fimmu.2021.730706