The lung's microbial flora regulates the brain's immune reactivity
In its vital function of providing the body with oxygen, the lung is connected to the outside environment by a large exchange surface. This boundary between lung tissue and outside air is inhabited by a specific microbial flora, the so-called lung microbiome. The exact function of this microbiome has been scarcely researched. A scientific team led by Profs. Alexander Flügel and Francesca Odoardi at the Institute for Neuroimmunology and Multiple Sclerosis Research, University Medical Center Göttingen, have shown a close relationship between the lung microbiome and the brain. The researchers found out that the lung microbiome regulates the activity of microglia, the "brain's immune cells." This newly ascertained lung-brain axis is significant for disease processes: the exact composition of the lung microbiome determines the susceptibility of developing an autoimmune inflammation in the brain such as occurs in multiple sclerosis. The experimental results of this work were published today, 23 February 2022, in the online edition of the journal Nature.
This research by the Göttingen scientists points to a new and unexpected functional connection between the lung and brain. This connection is regulated by the local lung microbiome, which apparently is constantly sending signals to the microglia, the brain's "immune cells."
"The microglia adapt their immunological response capacity according to these microbial signals and can therefore react quickly to impending threats" stated Dr. Leon Hosang, postdoc and Klaus Faber Fellow at the Institute for Neuroimmunology and Multiple Sclerosis Research and the publication's first author. "The lung microbiome can therefore be seen as an early warning system for the sensitive brain tissue," explained Francesca Odoardi, Heisenberg Professor and the publication's co-senior author. This can have consequences for our health: lung infections, therapeutic manipulations (antibiotic treatment), pollution and climate changes can all exert influence on the lung microbiome and thereby influence the immune reactivity of the brain.
"This new knowledge about the lung-brain axis has the potential for therapeutic implementation," stated Prof. Alexander Flügel, the publication's co-senior author. He continued: "For example, a dosage of probiotics or specific antibiotics could be used to influence the brain's immune reaction in a targeted fashion in order to treat not only multiple sclerosis but also diseases of the central nervous system in general where the immune activity of microglia plays a role."
Background information: Lung and brain
The lung can play an important role in disease processes in the brain. For example, lung infections and smoking increase the risk for developing multiple sclerosis. This disease belongs to the group of autoimmune disorders, where the immune system, in this specific case so-called T cells, attack the body's own brain tissue and cause lasting damage there which can lead to serious neurological deficits. Why and how it is the lung that should participate in regulating the brain's autoimmune processes has been a mystery up to now.
After all, it is the lung's main function to provide oxygen, for which its tissue forms an exchange area for the air we breathe in that has an area larger than 100 m2. It is true that this enormous contact area to the outside environment requires a specialized immune protection against dangerous invaders, e.g. viruses or bacteria, however the accepted understanding is that the body's main immune reactions take place above all in specialized immune organs such as the lymph nodes or spleen. In those organs, all the cells and soluble immune factors are present to start and regulate an immune answer not only to invaders but also to the body's own tissue.
Changes to the lung microbiome influences the brain's susceptibility to an autoimmune inflammation
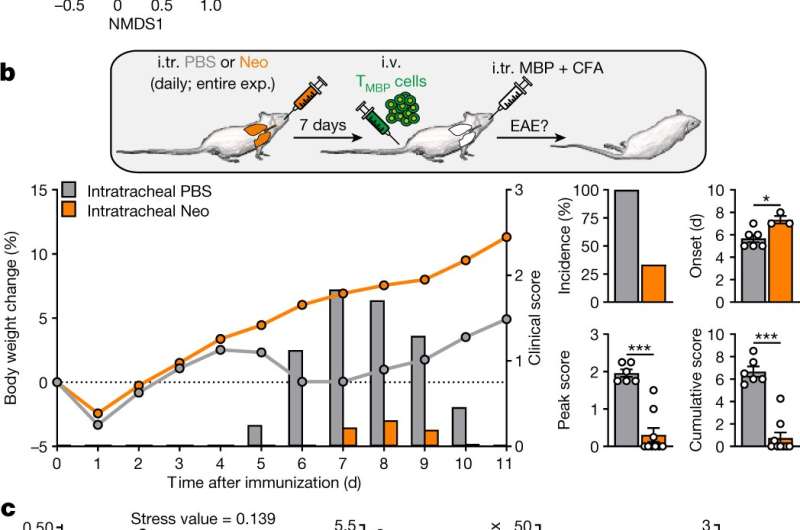
The researchers have now found surprising answers to the question of how the lung influences autoimmune reactions in the brain. They found out that the lung's bacterial colony on the contact areas between lung tissue and respiratory air, the so-called lung microbiome, plays a special role here. The human body is populated by bacteria on every contact surface to the outside world. While the composition of the gut and skin microbiomes are well understood, the lung microbiome has been comparatively little researched. Indeed, it was believed for a long time that the lung is sterile, in other words has no bacterial population at all. Actually, the lung does have a quantity of microbes, but compared to the gut this quantity is low (smaller by a factor of 108). It was all the more surprising for the Göttingen scientists when they found out that just a small manipulation of the microbial flora via a local application of a low dose of antibiotics was sufficient to strongly change the brain's susceptibility to developing an autoimmune disease.
Cell wall components of lung bacteria regulate microglia, the brain's immune surveillance
How does the lung microbiome carry out this influence on autoimmune disease in an organ as far away as the brain? The researchers pursued the answer to this question like detectives, where they investigated step-by-step at which locations the influence takes effect, which locations are affected and which bacterial signals exert influence on the cell regulation. They discovered that the point of attack was not in the lung or the other immune organs of the body, but rather directly in the brain tissue itself. There, upon an antibiotic-mediated manipulation of the lung microbiome, they encountered measurable changes in the brain's microglia, changes that could even be observed under the microscope. In comparison to other brain cells, microglia are small finely branching cells, which are constantly scanning their surroundings with their delicate cell processes for damage or danger by infectious agents, and upon finding it, just like the immune cells in other regions of the body, sound the alarm and summon immune help.
After the antibiotic dose in the lung, the microglia's branches were shorter and thicker, and the microglia reacted more weakly to inflammatory signals, resulting in a reduced recruitment of immune cells to the inflamed brain tissue. This provides a good explanation for the change in susceptibility to autoimmune inflammation. In a final step, the researchers looked for bacterial signals that could trigger such a "microglial paralysis." Vital clues were provided by analyses of the lung bacteria, carried out with help from scientists from the Institute for Microbiology, Faculty of Biology, University Medical Center Göttingen. These analyses indicated that those bacteria producing a specific cell wall component, namely lipopolysaccharide, accumulated more strongly in the lung tissue after the antibiotic dosage. The increased amount of lipopolysaccharide indeed triggered the observed microglial paralysis and autoimmune resistance. However, a reduction of lipopolysaccharide in the lung caused the opposite: the autoimmune disease intensified.
More information: Leon Hosang et al, The lung microbiome regulates brain autoimmunity, Nature (2022). DOI: 10.1038/s41586-022-04427-4