Peptide delays melanoma growth in animal trials
An article published in Scientific Reports describes a study demonstrating the effectiveness of a peptide developed by Brazilian scientists, called Rb4, in combating cancer progression in an animal model, especially malignant melanoma. The molecule holds promise for the treatment of drug-resistant tumors.
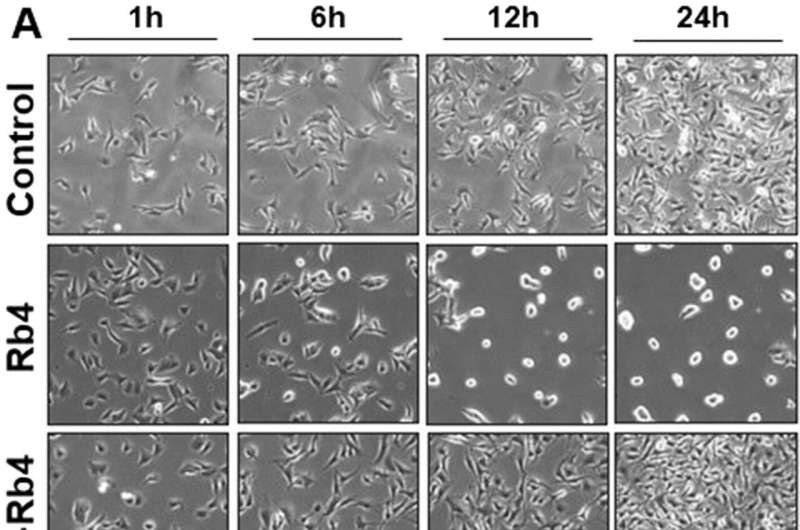
Preclinical in vitro and in vivo trials indicated that Rb4 triggers necrosis in murine melanoma cells, and inhibits the viability of human cancer cells. Tumor cells in the study lost the integrity of their plasma membranes, and mitochondria (energy-producing organelles) dilated even in the absence of chromatin condensation, a morphological hallmark of apoptosis. How this necrosis is triggered is still poorly understood, the researchers acknowledge.
In mice, the peptide reduced lung metastasis and slowed subcutaneous melanoma growth. The results suggest that Rb4 acts directly on tumors, inducing expression of two damage-associated molecular patterns (DAMPs), which trigger immunogenic melanoma cell death.
"We do basic science in a search for novel molecules. In this study Rb4, which is derived from proteolipid protein 2 [PLP2], displayed a preference for causing necrosis, a specific type of cell death, especially in melanoma, but how this necrosis occurs and develops isn't clear. The article discusses some aspects of the peptide's morphological composition and the final effects of contact with it," Fabrício Castro Machado, a co-author of the article, told Agência FAPESP.
With FAPESP's support, a group led by Luiz Rodolpho Travassos, an emeritus professor at UNIFESP, began conducting the research. The authors of the article pay tribute to Travassos, who died in 2020. He published more than 230 articles in leading scientific journals, many of them on studies of peptides and peptidases (enzymes that break down proteins into peptides and eventually into single amino acids) in infectious diseases and cancer.
"Professor Travassos identified several sequences of bioactive peptides, small molecules based on antibodies developed by ReceptaBio. Rb4 was also identified during this process of searching for novel molecules, although it isn't derived from antibodies. We have another, Rb9, which is at a more advanced research stage, with several publications and patents, but still at the preclinical stage," said Alice Santana Morais, a research and development analyst at ReceptaBio and corresponding author of the article.
In 2016, the scientists described the structure of Rb9 and its action mechanism as an inhibitor of melanoma cells. A more recent article published in 2020 showed that Rb9 acts as an immunomodulator and can be used to control tumor progression.
"Whether in academia or in companies like ReceptaBio, we need to combine efforts to conduct research. We're looking for partners to boost the drug development process, which is lengthy and painstaking, and requires discussion, details, and an exchange of experiences," Morais said.
Promising results
Novel cancer treatments developed in recent years include peptide-based chemotherapy. Peptides have received increasing attention not only because they can bind to the membranes of tumor cells, but also because they have low molecular weights, good cell tissue penetration, and low toxicity for normal tissue. They can be used as cell reagents, ligands, vaccines and carriers of cytotoxic drugs in peptide-alone therapy or peptide-conjugated materials.
In the study on Rb4's anti-tumor action, the group found that the peptide interfered with the morphology, replication and association of B16F10-Nex2 melanoma cells cultured in the laboratory. In contrast with controls, cells treated with Rb4 did not replicate and formed clusters, losing their natural morphology after incubation for at most 24 hours.
In addition, Rb4 reduced the number of lung metastatic nodules in a syngeneic melanoma model (involving tumor tissues from mice with the same genetic makeup). This result was detected after melanoma cells were injected intravenously into the mice. They were given five intraperitoneal injections of the peptide (300 micrograms per animal) on alternate days, delaying tumor growth by up to 40 days.
The survival rate of mice treated with Rb4 was significantly greater than that of the controls, increasing group survival by more than 25% and up to 10 days.
More information: Vera S. C. Maia et al, PLP2-derived peptide Rb4 triggers PARP-1-mediated necrotic death in murine melanoma cells, Scientific Reports (2022). DOI: 10.1038/s41598-022-06429-8
Natalia Girola et al, The Ig V H complementarity-determining region 3-containing Rb9 peptide, inhibits melanoma cells migration and invasion by interactions with Hsp90 and an adhesion G-protein coupled receptor, Peptides (2016). DOI: 10.1016/j.peptides.2016.08.006
Fabrício C. Machado et al, Immunomodulatory Protective Effects of Rb9 Cyclic-Peptide in a Metastatic Melanoma Setting and the Involvement of Dendritic Cells, Frontiers in Immunology (2020). DOI: 10.3389/fimmu.2019.03122