Breast cancer: New drug shrinks brain metastases
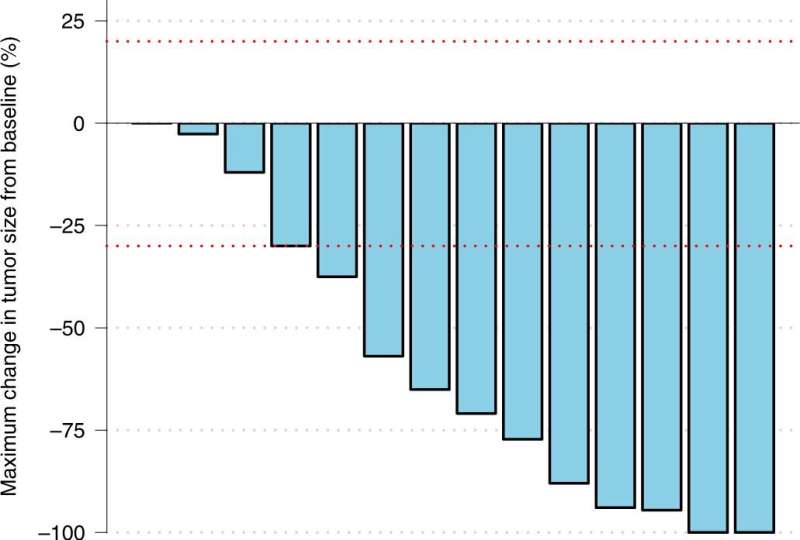
An Austrian study led by MedUni Vienna showed that active brain metastases in breast cancer patients are partially or even completely regressed by a novel class of drug. This is a chemical conjugate of an antibody and a chemotherapy drug that, according to current findings, opens up a completely new avenue in oncological research and targeted therapy. The study results were published in the journal Nature Medicine and are considered ground-breaking in the treatment of brain metastases, a dreaded complication of cancer.
The study included 14 women and one man with HER2-positive breast cancer and brain metastases, who were being treated by the Division of Oncology of MedUni Vienna and University Hospital Vienna. In the study, the Austrian research team led by Matthias Preusser and Rupert Bartsch (Division of Oncology within the Department of Medicine I of MedUni Vienna and University Hospital Vienna) investigated the agent trastzumab deruxtecan (T-Dxd) as a possible new therapeutic approach in cases where breast cancer spreads to the brain.
The findings were as follows: T-Dxd shrank the metastases in 73.3% of patients and made them completely disappear from brain scans in two out of 15 patients (13.3%). In addition to this extremely positive result, the researchers also found that the drug was well tolerated: there was no deterioration in the participants' brain function or quality-of-life during the treatment period. Moreover, T-Dxd is already approved within the EU and other parts of the world: "It can therefore be used immediately for the treatment of breast cancer patients with brain metastases in specialized oncology units in Austria and internationally," points out study leader Matthias Preusser.
50% develop brain metastases
With more than 5,000 new cases per year, breast cancer is the most common cancer in women in Austria. This type of cancer also affects less than 1% of men. 15% of breast cancer patients have HER2-positive breast cancer. In this aggressive form of cancer, HER 2 (Human Epidermal Receptors) act as docking sites for growth factors that drive the cancer cell to divide and thus to grow and metastasize. Cancer metastasizes to the brain in up to 50% of patients with metastatic HER2-positive breast cancer.
Antibody-chemotherapy drug conjugate
T-Dxd was approved by the European Medicines Agency (EMA) in 2021 for the treatment of unresectable or metastatic HER2-positive breast cancer. It is a chemical conjugate of an antibody against HER2 (trastuzumab) and a chemotherapy drug (deruxtecan). Until now, it was not known whether the novel conjugate could be effective in active brain metastases.
On the basis of the recent study results, further investigations are now planned into the novel class of drug: "Our findings open up entirely new avenues for clinical research and the treatment of brain metastases in breast cancer—and possibly other types of cancer as well," says Matthias Preusser, looking to the future of cancer treatment.
More information: Rupert Bartsch et al, Trastuzumab deruxtecan in HER2-positive breast cancer with brain metastases: a single-arm, phase 2 trial, Nature Medicine (2022). DOI: 10.1038/s41591-022-01935-8


















