August 18, 2022 feature
Study unveils a global dysregulation of dopamine in mice with chronic inflammatory pain
Over the past decades, many neuroscientists have investigated how the mammalian brain modulates pain, trying to uncover the neural mechanisms underpinning chronic pain. Chronic pain is pain in specific parts of the body, that persists for long periods of time, including arthritis, back pain, neck pain or other forms of pain.
While past studies have led to important discoveries about pain modulation, many questions remain unanswered. For instance, while neuroscientists have identified some of the brain regions involved in modulating pain, the way in which pain-related cortical regions receiving dopaminergic inputs process pain is poorly understood.
Researchers at University of Toronto Mississauga have recently carried out a study exploring the effects of chronic inflammatory pain on the brain and affective state of mice. Their findings, published in Elsevier's Neuroscience journal, suggest that inflammatory pain can alter the process through which dopaminergic neurons modulate excitatory synapses, causing them to become hypersensitive and anxious.
"There are several unknowns about chronic pain, but one of the most critical areas that needs attention are the brain mechanisms underlying the modulation of pain responses," Loren J. Martin, one of the researchers who carried out the study, told Medical Xpress.
"Our work was inspired by previous investigations that have examined cortical brain region involvement in the development and maintenance of chronic pain."
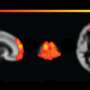
Initially, Martin and his colleagues tried to determine whether chronic inflammatory pain altered the excitability of the anterior cingulate cortex (ACC) in the mice brain. The AAC is an important brain region known to play a role in the expression of emotions, the allocation of attention, and mood regulation. In the context of pain, this brain region can impact how pain is perceived and expressed on an emotional level.

"Dopamine, a predominant neurotransmitter in the brain, is often dysregulated in diseases such as Parkinson's disease, schizophrenia and depression," Martin explained. "What is under appreciated in these disorders—except for depression—is that they have underlying pain phenotypes that are often ignored but suggest that overall changes in dopaminergic signaling importantly modify pain perception."
The recent study by Martin and his colleagues builds on past findings that hinted at the possible role of dopamine in the perception of pain. Their goal was to determine whether alterations in dopaminergic transmission within the AAC contributed to chronic inflammatory pain.
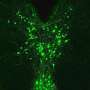
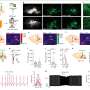
"We primarily used synaptic physiology—electrophysiology—to examine whether transmission between neurons was altered in the ACC during inflammatory pain," Martin said. "We also used HPLC to measure dopamine levels in the ACC of mice with inflammatory pain. Overall, we find that neurons of the ACC were potentiated and that dopamine, which normally inhibits transmission in the ACC was not as efficient at inhibiting neurons during the inflammatory state."
The experiments carried out by this team of researchers yielded numerous interesting findings. Most notably, they unveiled a global dysregulation of dopamine in mice experiencing chronic pain caused by inflammation in their hind paws. The team found that the mice also exhibited hypersensitivity and transient anxiety.
The neurotransmitter dopamine is known to be involved in reward or pleasure-seeking behavior, memory, movement and motivation. The impact of dopamine dysregulation on the mood alterations associated with chronic pain, however, has not yet been widely studied.
The recent work by Martin and his colleagues could inspire other teams to investigate the link between chronic inflammatory pain and dopaminergic transmission in the AAC. This could ultimately help to better understand the neural underpinnings of emotional issues associated with human chronic pain.
"We will now most likely examine the connectivity of dopamine neurons to and from the ACC as a future direction to understand how these neurons control pain and its emotional aspects," Martin added.
More information: Soroush Darvish-Ghane et al, Inflammatory Pain Alters Dopaminergic Modulation of Excitatory Synapses in the Anterior Cingulate Cortex of Mice, Neuroscience (2022). DOI: 10.1016/j.neuroscience.2022.07.010
© 2022 Science X Network