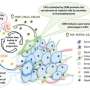
Researchers uncover how breast cancer cells become resistant to therapy
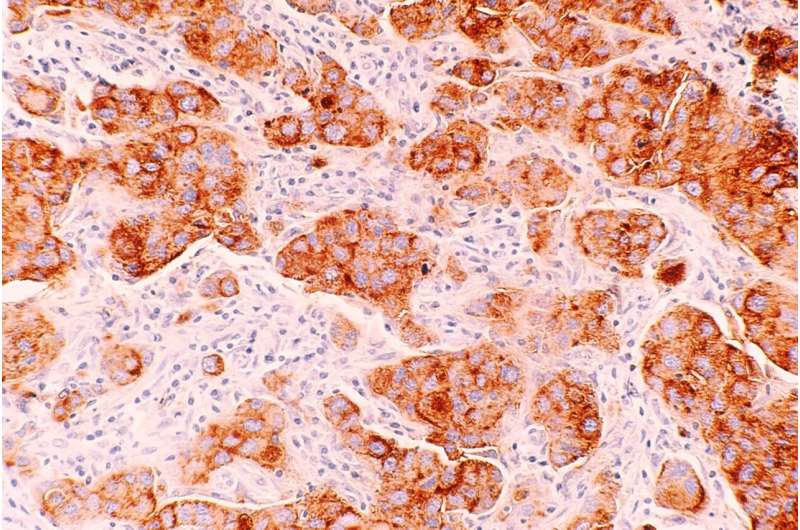
About one-fourth of recurrent estrogen receptor-positive (ER+) breast cancers lose ER expression, which renders them resistant to endocrine therapy and able to grow uncontrolled. A team of researchers at Baylor College of Medicine has investigated how these cells lose their ER, and in the current study published in the Proceedings of the National Academy of Sciences, they reveal a mechanism that not only explains the process but also offers possibilities to overcome it.
"For years, our goal has been to tease out the complex puzzle of breast cancer progression to understand how the players interact with each other to confer resistance to therapy and persistent growth," said corresponding author Dr. Weei-Chin Lin, professor of medicine—hematology and oncology and of molecular and cellular biology at Baylor. "Our goal is to overcome this hurdle to restore ER receptor expression in these cancers so they become susceptible to therapy again, giving patients a better chance for recovery."
How breast cancer cells lose their ER
Two cellular proteins known as 14-3-3τ and ERα36 have been previously implicated in the development of breast cancer resistance to endocrine therapy.
"Working with a mouse model of human ER+ breast cancer, we were surprised to find that over-expressing 14-3-3τ in these tumors led to all the cancer cells becoming ER-negative (ER-)," said Lin, a member of the Dan L Duncan Comprehensive Cancer Center. "I still remember the day I saw the data. The change was dramatic—all the tumors had lost their ER."
Studying the mechanism in animal models would be labor intensive, time consuming and expensive, so the researchers developed an alternative model. First author Lidija A. Wilhelms Garan, a student in Baylor's Cancer and Cell Biology Graduate Program working in the Lin lab, developed a spheroid model of human breast cancer cells that mimics the progression from ER+ to ER- and provides a very useful experimental tool for future investigation.
"In a patient, a breast tumor can take years to progress from ER+ to ER-, in our animal model it takes several months but in our spheroid model it switches from ER+ to ER- in 1 to 2 weeks," Garan said.
In the lab spheroid model the team found that once 14-3-3τ is over-expressed in cancer cells under the right conditions, the cells will increase their levels of ERα36 and this is followed by ER loss.
"Other molecular players, such as AKT and GATA3, also are required," Garan said. "Importantly, we also found that factors produced by the tumor microenvironment, which includes fibroblasts and immune cells that are part of the tumor mass and cross talk with the cancer cells, also are essential for the progression from ER+ to ER-."
"We knew that 14-3-3τ, ERα36, AKT and GATA3 were the key players involved in turning ER+ breast cancer cells into ER- cells. Here we have determined how they functionally interact with each other, laying out a map of the road that leads to ER loss," Lin said. "I am very excited that with our spheroid breast cancer model we now have a valuable tool to study not only the cellular changes involved in breast cancer progression but also to test drugs for their ability to inhibit the process that leads to ER loss."
"The protein 14-3-3τ is overexpressed in about 60% of breast cancers. Not all patients that have high 14-3-3τ will lose the ER, but for those who do, our findings may one day help restore their tumors to a therapy-sensitive state," Garan said. "The translational aspect of this research has always been close to my heart—to bring discoveries to the clinic and improve people's lives."
Yang Xiao at Baylor College of Medicine also was an author of this work.
More information: Lidija A. Wilhelms Garan et al, 14-3-3τ drives estrogen receptor loss via ERα36 induction and GATA3 inhibition in breast cancer, Proceedings of the National Academy of Sciences (2022). DOI: 10.1073/pnas.220921111. www.pnas.org/doi/10.1073/pnas.2209211119