This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Unraveling key determinant of successful therapeutic vaccination against chronic hepatitis B
Hepatitis B virus (HBV) infections remain a major global health problem—according to the World Health Organization there are around 300 million HBV carriers worldwide. Current treatments rarely succeed in curing the infection.
At present, therapeutic vaccination represents a promising approach to cure HBV. But what are the key molecular factors responsible for its efficacy? A team led by Ulrike Protzer and Anna Kosinska from the Institute of Virology at Helmholtz Munich, the Technical University of Munich (TUM) and the German Center for Infection Research (DZIF) has determined that the success of therapeutic hepatitis B vaccination depends on efficient priming of HBV-specific CD4 T cells.
According to the World Health Organization (WHO), more than 3% of the world population suffers from chronic hepatitis B virus (HBV) infection which is the leading cause of liver cirrhosis and liver cancer. Current treatments are rarely able to achieve a cure in these patients. Therapeutic vaccines (vaccines that stimulate the immune system to cure a disease) represent a promising approach to cure HBV but have shown limited clinical efficacy so far.
During the last two decades, numerous attempts have been made to develop effective therapeutic hepatitis B vaccines. However, only few of these vaccine candidates entered clinical trials and none of them showed antiviral efficacy in chronic hepatitis B patients up to date. This highlights the necessity of dissecting the factors essential for a successful therapeutic vaccination and target them by rational vaccine design.
Efficient CD4 T-cell activation determines the success of therapeutic vaccination
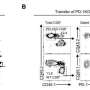
A team of researchers around Ulrike Protzer and Anna Kosinska are currently investigating TherVacB, a novel heterologous prime-boost therapeutic hepatitis B vaccine. In heterologous prime-boost vaccines, the same antigens are presented in different forms to the immune system. In TherVacB, proteins are used to prime the immune response, while for boost vaccination, the antigens are delivered by a viral vector.
The benefit of heterologous prime-boost vaccination is a stronger and longer-lasting immunity. However, optimization of the prime- and boost vaccination strategies requires a thorough understanding of how which vaccine component activates the different cells of the immune system, like T- or B cells.
In this study, first author Jinpeng Su and her colleagues demonstrated that the success of therapeutic hepatitis B vaccination depends on efficient priming of HBV-specific CD4 T cells. CD4 T cells are a specific group of lymphocytes, that carry the CD4 molecule on their surface and coordinate the immune defense against foreign pathogens. The researchers showed, that the depletion of CD4 T cells led to a complete loss of TherVacB-induced antiviral efficacy in preclinical mouse models. Moreover, applying an optimal protein vaccine formulation during prime immunization allowed to properly activate CD4 T cells, and laid the foundation for boost vaccination to control HBV infection.
The study, published in the Journal of Hepatology, deciphers the effect of the individual vaccine components and provides the first direct evidence that efficient activation of HBV-specific CD4 T cells is indispensable for initiating immune control of HBV. It also determines which components are required for a successful therapeutic hepatitis B vaccination and gives important insights into the rational design of therapeutic vaccines for the urgently needed clinical application.
Impact for vaccine development
The researchers' work further confirms the effect and explains the mode of action of TherVacB, which will be evaluated in a clinical trial, starting in 2023. They therewith greatly support the vision of overcoming HBV-specific immune tolerance and restoring the antiviral B-cell and T-cell response through therapeutic vaccination to cure HBV once and for all.
Further, this study confirms and expands the textbook knowledge in a concrete case and underlines the importance of the prime vaccination to nudge the immune system in the desired direction. It thus serves to develop or improve therapeutic vaccines against chronic hepatitis B—and perhaps other chronic or malignant diseases—and hopefully helps to accelerate the progress towards a cure for chronic hepatitis B, which will be a great benefit for millions of hepatitis B patients worldwide.
More information: Jinpeng Su et al, Activation of CD4 T cells during prime immunization determines the success of a therapeutic hepatitis B vaccine in HBV-carrier mouse models, Journal of Hepatology (2023). DOI: 10.1016/j.jhep.2022.12.013